Research Article
Antimicrobial Activity and Phytochemical Screening of Sarcocolla Gum Resin
Department of Biology, College of Science, Imam Abdulrahman Bin Faisal University, P.O. Box 1982-31441, Dammam, Saudi Arabia
LiveDNA: 966.22342
The problem of microbial resistance to antimicrobial drugs is being increasingly recognized. Vashishtha1 reported that only one antibacterial medicine ‘doripenem’ has been approved in the USA since, 2006. Recently Frieden2 reported that the development and production of antibiotics has significantly decreased since, 2000. Secondary plant metabolites may be an ideal source of new antimicrobial agents due to their ability to inhibit the growth of diverse microbes, including multidrug resistant bacteria3,4.
Sarcocolla is a gum resin that can be extracted from the Astragalus sarcocolla tree that belongs to the Fabaceae, family. Astragalus is a very large genus that includes 2326 species5. Sarcocolla has different names in different countries and is known as: Sarcocolla in English, Anzerut, Gujar, Guzhad and La-ie in Urdu, Rudanti in Ayurvedic, Anzaroot and Kohal Kirmaani (Gum) in Unani and Anzaroot in Arabic6,7. Sarcocolla has been used since ancient times as a medicine due to its ability to its properties as an antirheumatic, anthelmintic, aperient and emollient7,8.
Sarcocolla gum resin is one of the most famous gum resins in Saudi Arabia, especially in the Northern region and is used to treat flatulence, wounds, burns and to prevent wound bleeding. Also it is used in children treatments, where it is usually added to hot drinks in small quantities to prevent various winter diseases according to folk heritage. Despite its lengthy use, Sarcocolla gum resin has not been studied as an antimicrobial agent. Therefore, researchers investigated the ability of Sarcocolla gum resin extract to inhibit the growth of pathogenic Gram-positive bacteria, Gram-negative bacteria and yeast. The phytochemicals of Sarcocolla gum resin were also screened.
Plant material: Sarcocolla "Anzaroot" gum resin was purchased from a folk medicine market in Dammam City, Saudi Arabia, in July, 2016. The plant material was transferred to the microbiology laboratory and kept in refrigerator at 4°C until use.
Plant extracts preparation: Sarcocolla gum resin was cleaned and finely ground to a powder using a blender. Five, 10 and 15 g of ground Sarcocolla were added separately to 500 mL Erlenmeyer flasks. Twenty-five milliliter of distilled water and ethanol (80%) was added to each flask separately. The final Sarcocolla concentrations (g mL–1) were 20, 40 and 60%, respectively. The Erlenmeyer flasks were placed in a shaker at 300 rpm at 20°C for 3 days. The mixtures were filtered using Whatman No. 1 filter paper followed by bacterial filters. The filtrates were stored at 4°C until use.
Qualitative phytochemical analysis: Phytochemical screening was qualitatively assessed using standard procedures9. To detect alkaloids, 2 mL of plant extract was added to 10 mL of HCl (1%) and the mixture was transferred to a water bath at 60°C for 30 min. The mixture was then filtered using Whatman No. 1 filter paper. Three drops of Wager’s reagent was added to 2 mL of the filtrate. A reddish-brown color was considered a positive result. To detect sterols, 2 mL of plant extract was added to two drops of concentrated Sulfuric acid and formation of a reddish-brown ring at the interface was considered a positive result. To detect tannins, 10 mL of distilled water was added to 0.5 mL of plant extract and mixed thoroughly. Then 0.5 mL of lead acetate (1%) was added and formation of a gelatin precipitate was considered a positive result. To detect flavonoids, 2 mL of sodium hydroxide (2%) was added to 1 mL of plant extract. If a yellow color appeared then 3 drops of hydraulic acid (4%) were added. Disappearance of the yellow color was considered a positive result. To detect terpenoids, 3 mL of sulfuric acid was added to 2 mL of plant extract and the mixture was heated using water bath at 60°C for 2 min. Appearance of a greyish color was considered a positive result. To detect saponins, 5 mL of plant extract was transferred to a test tube and shaken thoroughly for 5 min using a vortex mixer. Formation of foam (about 2 cm layer) that remained for 30 min was considered a positive result.
Test microorganisms: Five bacterial species and yeast were used. The test microorganisms included: Three Gram-negative bacteria (Escherichia coli ATCC 35218, Pseudomonas aeruginosa ATCC 27853 and Enterobacter aerogenes), two Gram-positive bacteria (Staphylococcus aureus ATCC 29213 and Bacillus cereus) and one yeast Stephanoascus ciferrii. All Gram-negative bacteria and S. aureus ATCC 29213 were obtained from King Fahd Hospital, AlKhobar, Saudi Arabia. B. cereus and S. ciferrii were obtained from the Biology Department, College of Science, Imam Abdulrahman Bin Faisal University.
Screening of antimicrobial activities: Well diffusion assays were used according to the National Committee for Clinical Laboratory Standards10. One milliliter of bacterial culture age 18-24 h (standard inoculum 1-2×108 CFU mL–1 0.5 McFarland standard) were add to petri plates and 15 mL nutrient agar was poured into the plate. After the media solidified, holes were made using a 5 mm cork borer and each hole was filled with 50 μL of plant extract. Inoculated agar plates were placed in a refrigerator for 1 h to allow diffusion of the plant extract then plates were incubated overnight at 37°C. The same solvents (water and ethanol) without plant extract were used as negative controls and bacterial antibiotic (streptomycin 10 mcg) and fungal antibiotic (nystatin 100 mg) were used as a positive controls. Zones of inhibition around each well were recorded in millimeters.
Determination of minimum inhibitory concentration (MIC): MICs of the plant extracts were determined using two-fold dilution method as described by Omura et al.11. The 96-well microtitre plates were used and MICs values were read after overnight incubation at 37°C, using microtitre plate reader at a wavelength of 600 nm. All experiments were conducted in triplicate.
Statistical analysis: The impact of solvents and their concentrations on microbial growth was analyzed at p<0.01 using one-way ANOVA through SPSS version 17.0, Chicago, USA12.
Results of phytochemical analysis of Sarcocolla gum resin indicated the presence of alkaloids, flavonoids, sterols, terpenoids and tannins in both solvents. Whereas saponins appeared only in water extract (Table 1). All bioactive compounds possess inhibitory properties toward microbes.
Results shown in Table 2 demonstrated the effect of Sarcocolla gum resin extracts on the test microorganisms. Most of test microorganisms were inhibited by high concentrations of water and ethanol extract 40-60%. In contrast, water extracts inhibited the growth S. aureus ATCC 29213 at all concentrations. However, ethanol extracts did not inhibit the growth of this bacterium.
In addition, the MIC of Sarcocolla gum resin extract had a remarkable inhibitory effect (Table 3). The ethanol extract showed the highest inhibitory effect on P. aeruginosa and S. ciferrii at MIC 12.5 μg mL–1, whereas water extract had the highest inhibitory effect on the growth of P. aeruginosa ATCC 27853, S. aureus ATCC 29213 and S. ciferrii at MIC 12.5 μg mL–1.
| Table 1: | Photochemicals detected in the 60% concentration extract of Sarcocolla gum resin and their mechanism of action toward microbes |
 | |
| +: Exist, -: Not exist | |
| Table 2: | Antimicrobial activity of Sarcocolla gum resin extracts measured using the well diffusion method |
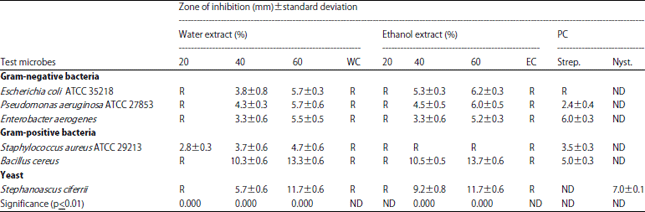 | |
| R: Resistant, WC: Water negative control, EC: Ethanol negative control, PC: Positive control (streptomycin 10 mcg and nystatin 100 mcg) , ND: Not Identify | |
| Table 3: | Minimal inhibitory concentration mg mL–1 (MIC) of Sarcocolla gum resin extract |
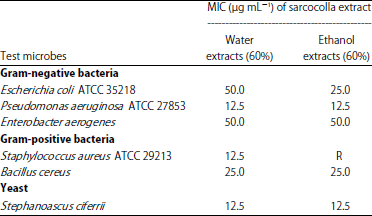 | |
| R: Resistant | |
Preliminary phytochemical screening of Sarcocolla gum resin showed the richness of this plant material with many secondary products like alkaloids, flavonoids, saponins, sterols, terpenoids and tannins. This finding agreed with several studies showing that plants contain secondary products2,22. All secondary metabolites found in Sarcocolla gum resin are harmful to microorganisms in different ways as previously described by several researchers listed in Table 1.
The results of this study agree, in part, with other preliminary phytochemical studies of Astragalus species. Balachandar et al.23 found that flavonoids, alkaloids, terpenoids and saponins appeared in both methanol and ethanol extracts of A. membranaceus. Also, phytochemical analysis by Jahangir et al.24 found that tannins, alkaloids, flavonoids, terpenoids, phenolics, sugars and saponins were present in methanolic, chloroform, n-hexane, butanol and ethyl acetate, extracts. Moreover, Jaradat et al.25 found that A. pelecinus contains glycosides, carbohydrates, proteins, phenolic tannins, flavonoids, alkaloids and terpenoids in ethanol, water, acetone and methanol extracts. In phytochemical analysis of A. maximus extract, Ghasemian-Yadegari et al.26 identified three flavonoids (formononetin, liquiritigenin, isoquercitrin) and one acylated cycloartane-type saponin, astragaloside I.
In agreement with several other publications27,28, researchers found a positive correlation between the inhibitory activity of water and ethanol extracts and their concentration, which may be due to the increased concentration of effective secondary metabolites. Moreover, the ability of the plant extract to inhibit microbial growth was significantly affected by the solvent type as previously reported by Wendakoon et al.29 and Ababutain30.
The results of this study showed that Sarcocolla gum resin extract possesses broad spectrum antimicrobial activity against both Gram-positive and Gram-negative bacteria, despite differences in cell wall structure between these two groups. Alternatively, it has been reported that Gram-negative bacteria are more resistant to external impacts than are Gram-positive bacteria31.
Sarcocolla gum resin extract also strongly inhibited the growth of tested yeast. The water extracts exhibited high MIC values of ranging 12.5-25 μg mL–1 against P. aeruginosa ATCC 27853, S. aureus ATCC 29213, B. cereus and S. ciferrii. Ethanol extracts exhibited high MIC values at 12.5 μg mL–1 against P. aeruginosa ATCC 27853 and S. ciferrii. This result is similar to research conducted on other species of Astragalus, such as the study by Jaradat et al.25, who found that aqueous extracts of A. pelecinus exhibited antimicrobial activity against S. aureus, MRSA, E. coli, P. aeruginosa and C. albicans. Methanol extract possessed antimicrobial activity against S. aureus, MRSA and E. coli. Moreover, Labed et al.32 found that the chloroform extract of A. armatus exhibited high antibacterial activity against S. aureus, E. coli and P. aeruginosa with MIC values of 80 μg mL–1.
The current study clearly showed that Sarcocolla gum resin possesses bioactive compounds that affect the growth of pathogenic microorganisms. Therefore, the use of such plant materials as a microbial therapeutic provides an opportunity to develop new antibiotics. Nevertheless, toxicity studies are required to ensure the safety of these plant materials. Since the solvent type has a direct effect on the inhibitory activity of plant extracts against microbes, testing other types of solvents will be required.
The current study revealed that Sarcocolla gum resin extracted from A. sarcocolla possessed high antimicrobial activity that depends on the solvent type, concentration of plant extract and microbial type. These results provide a possible new source of antimicrobials that may be useful in the manufacture of antibiotics.
This study demonstrated the antimicrobial activity of Sarcocolla gum resin against Gram-positive and Gram-negative bacteria and yeast that may be beneficial for the manufacture of new antibiotics. This is the first report of antimicrobial activity of Sarcocolla gum resin. Also, this study demonstrates that the inhibitory potential of plant extracts can be significantly affected by solvent type and plant extract concentration. Thus, conditions must be optimized to increase the inhibitory activity of plant extracts.
The author would like to thank the research units at Al Rayyan Campus-College of Science- Imam Abdulrahman Bin Faisal University for providing space and devices that were required for these experiments. I also would like to thank Dr. Ahmed Alsayyah, Dr. Reem AlJindan and Mrs. Nouf Alromaili for providing us with the tested bacteria.