Research Article
Effect of Environmental Factors on Cyanobacterial Abundance and Cyanotoxins Production in Natural and Drinking Water, Bangladesh
Department of Marine Biology, Faculty of Marine Sciences, King Abdulaziz University, P.O. Box 80207, Jeddah, 21589, Saudi Arabia
Hisham S. Khomayis
Department of Marine Biology, Faculty of Marine Sciences, King Abdulaziz University, P.O. Box 80207, Jeddah, 21589, Saudi Arabia
Salim Marzoog Al-Harbi
Department of Marine Biology, Faculty of Marine Sciences, King Abdulaziz University, P.O. Box 80207, Jeddah, 21589, Saudi Arabia
Mahfuzul Haque
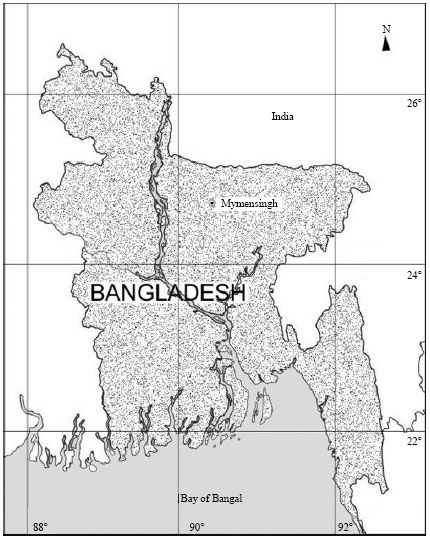
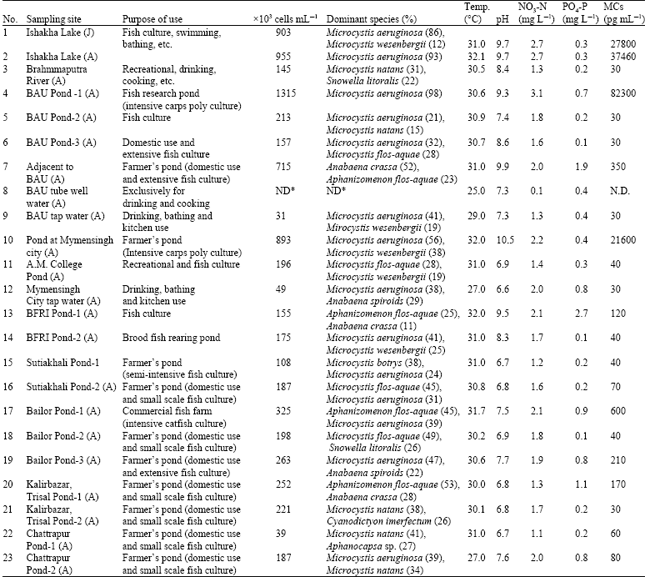
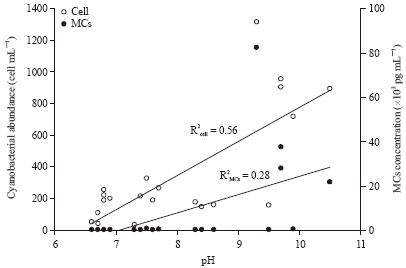
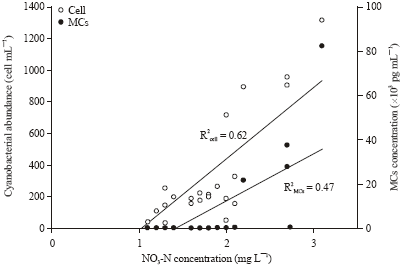
Department of Fisheries Management, Faculty of Fisheries, Bangladesh Agricultural University, Mymensingh, Bangladesh
Saleha Khan
Department of Fisheries Management, Faculty of Fisheries, Bangladesh Agricultural University, Mymensingh, Bangladesh