Research Article
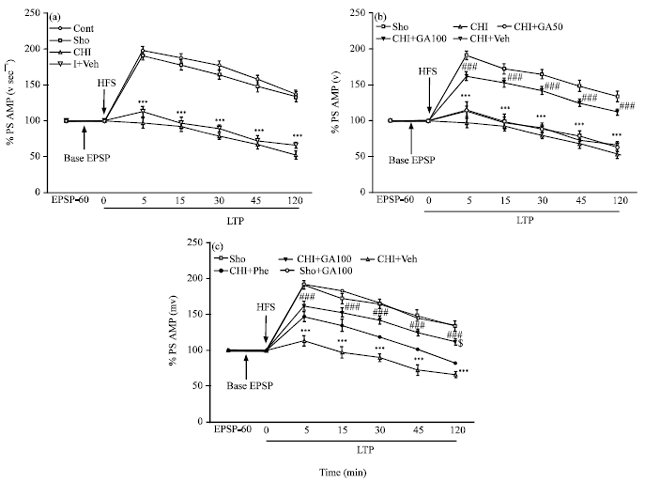
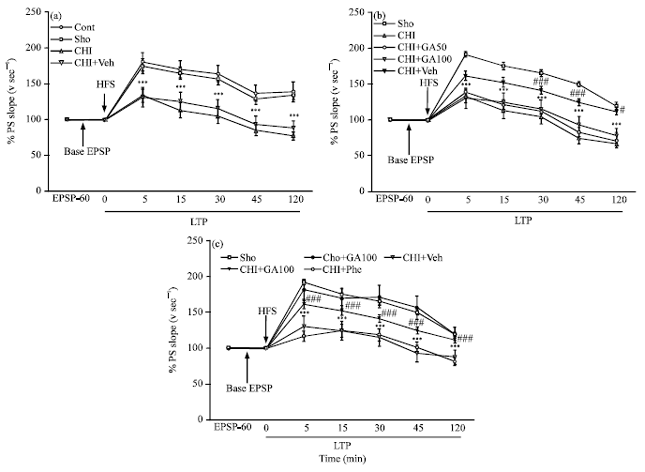
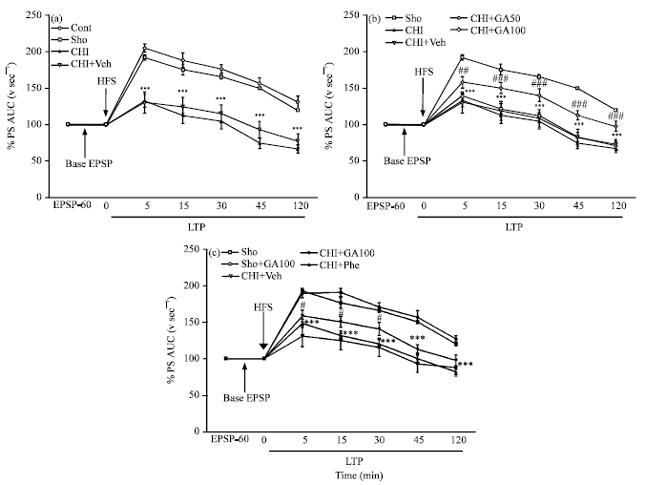
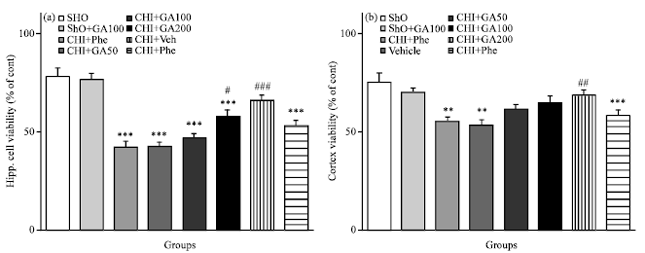
Gallic Acid Improves Cognitive, Hippocampal Long-term Potentiation Deficits and Brain Damage Induced by Chronic Cerebral Hypoperfusion in Rats
Ahvaz Physiology Research Center, Ahvaz Jundishpur University of Medical Sciences, Ahvaz, Iran
H. Fathimoghaddam
Department of Physiology, Ahvaz Jundishpur University of Medical Sciences, Ahvaz, Iran
S.M.T. Mansouri
Department of Pharmacology, Ahvaz Jundishpur University of Medical Sciences, Ahvaz, Iran
M. Shahrani Korrani
Medical Plant Research Center and Cellular, Molecular Research Center of Shahrekord University of Medical Sciences, Shahrekord, Iran
G. Saki
Department of Anatomical Sciences, School of Medical Sciences, Ahvaz Jundishpur University of Medical Sciences, Ahvaz, Iran
Y. Farbood
Medicinal Plant Research Center, Ahvaz Jundishpur University of Medical Sciences, Ahvaz, Iran