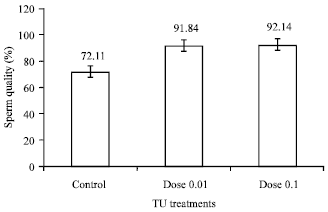
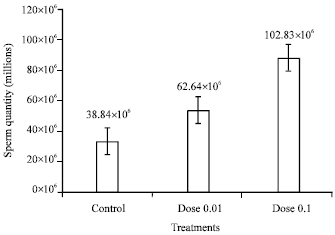
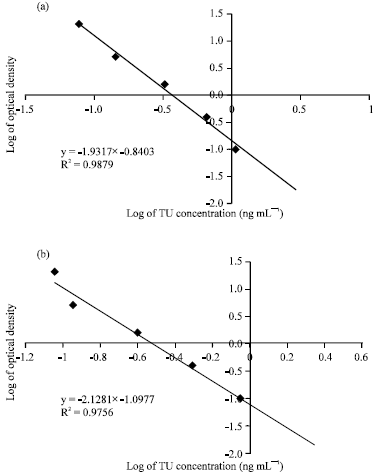
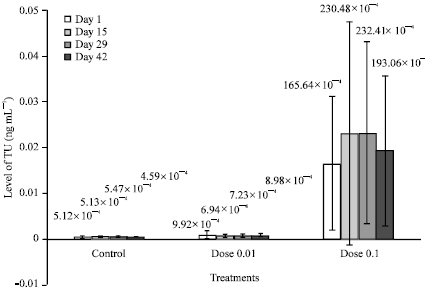
Research Article
Effect of Testosterone Undecanoate Hormone on Sperm and its Level in the Hemolymph of Male Mud Spiny Lobster, Panulirus polyphagus
Institute of Tropical Aquaculture, Universiti Malaysia Terengganu, 21030, Kuala Terengganu, Terengganu, Malaysia
J. Safiah
Institute of Tropical Aquaculture, Universiti Malaysia Terengganu, 21030, Kuala Terengganu, Terengganu, Malaysia
A.B. Abol-Munafi
School of Fisheries and Aquaculture Sciences, Universiti Malaysia Terengganu, 21030, Kuala Terengganu, Malaysia
M. Ikhwanuddin
Institute of Tropical Aquaculture, Universiti Malaysia Terengganu, 21030, Kuala Terengganu, Terengganu, Malaysia