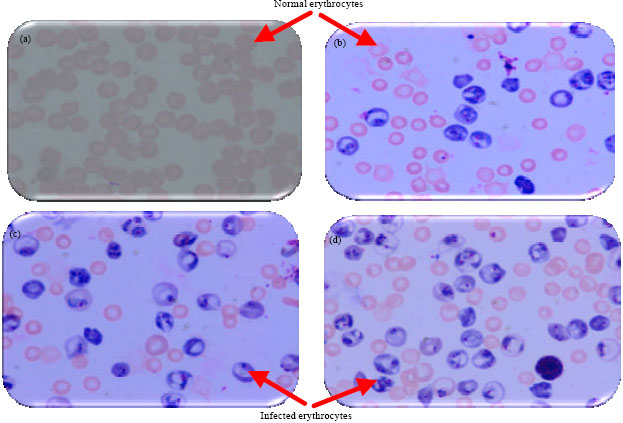
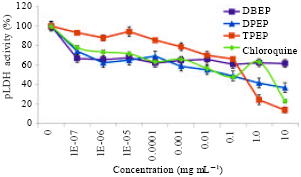
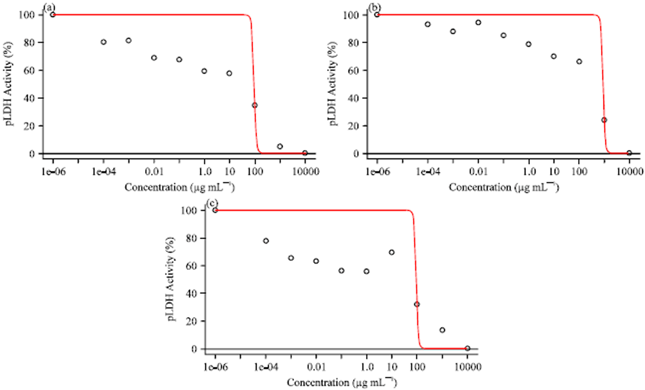
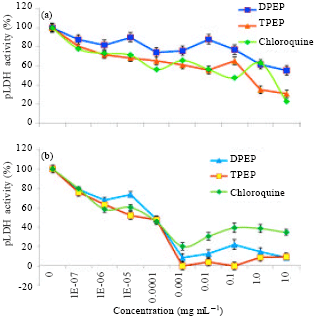
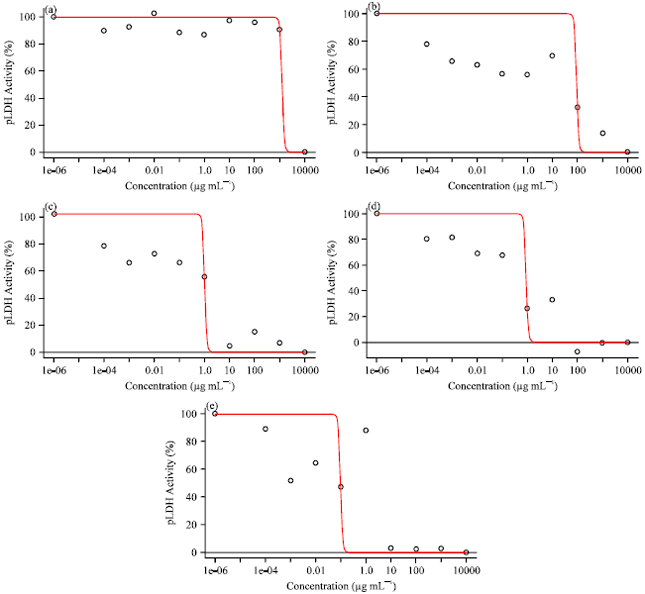
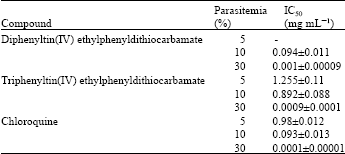
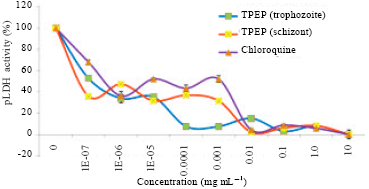
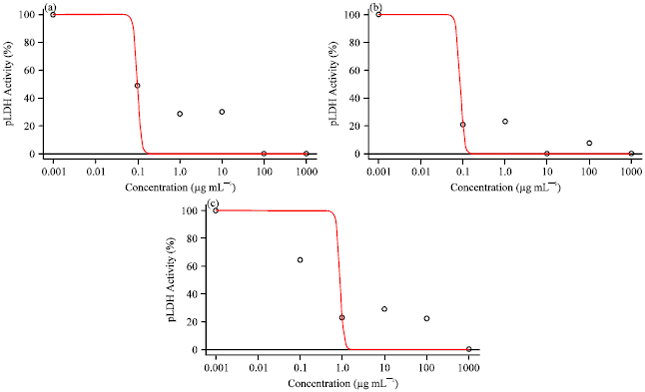
Research Article
Evaluation of the Ex vivo Antimalarial Activity of Organotin (IV) Ethylphenyldithiocarbamate on Erythrocytes Infected With Plasmodium berghei Nk 65
Programme Environmental Health and Industrial Safety, University Kebangsaan Malaysia, Jalan Raja Muda Abdul Aziz, 50300, Kuala Lumpur, Malaysia
Hafizah Jumat
Programme Environmental Health and Industrial Safety, University Kebangsaan Malaysia, Jalan Raja Muda Abdul Aziz, 50300, Kuala Lumpur, Malaysia
Shafariatul Akmar Ishak
Programme of Biomedical Science, Faculty of Health Sciences, University Kebangsaan Malaysia, Jalan Raja Muda Abdul Aziz, 50300, Kuala Lumpur, Malaysia
Nurul Farahana Kamaludin
Programme of Biomedical Science, Faculty of Health Sciences, University Kebangsaan Malaysia, Jalan Raja Muda Abdul Aziz, 50300, Kuala Lumpur, Malaysia