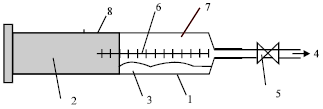
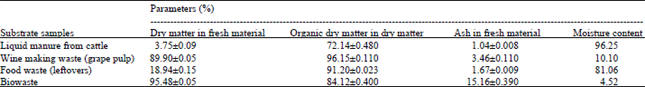
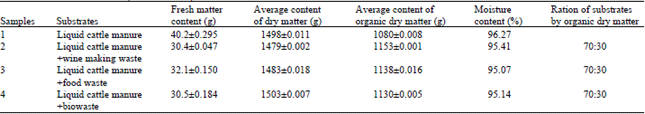
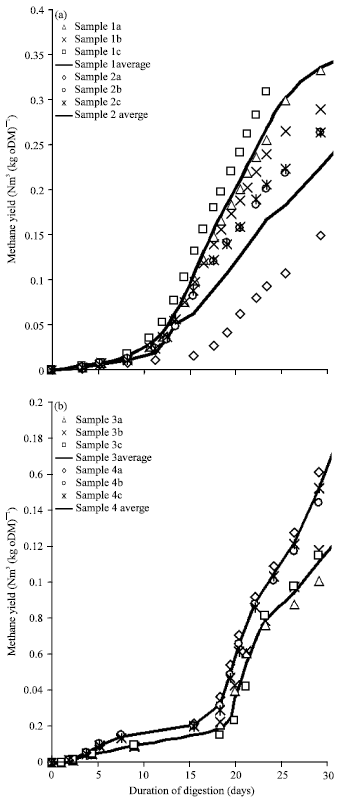
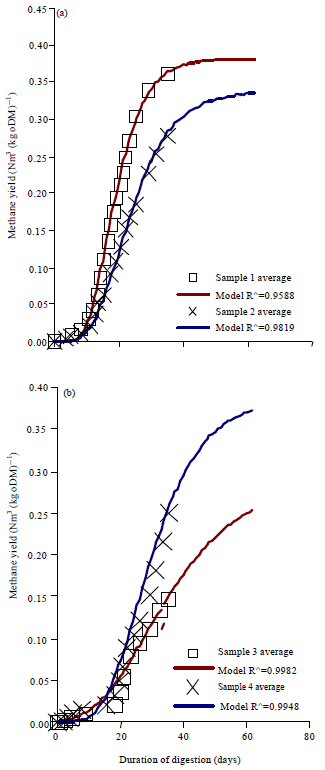
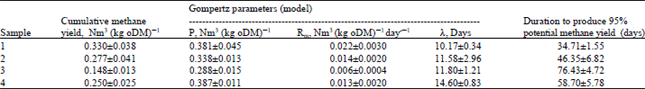
Research Article
The Kinetics of Methane Production from Co-digestion of Cattle Manure
Department of Biotechnology, M. Auezov South Kazakhstan State University, Shymkent, Kazakhstan
K.U. Korazbekova
Department of Biotechnology, M. Auezov South Kazakhstan State University, Shymkent, Kazakhstan
K.M. Lakhanova
Department of Biotechnology, M. Auezov South Kazakhstan State University, Shymkent, Kazakhstan