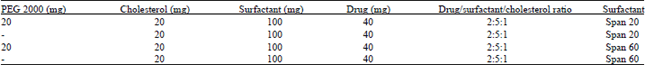
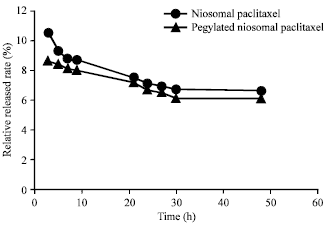
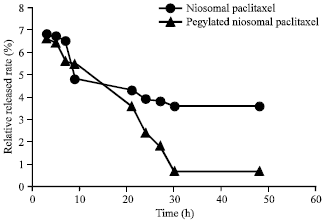
Research Article
Paclitaxel Loaded Niosome Nanoparticle Formulation Prepared via Reverse Phase Evaporation Method: An in vitro Evaluation
Department of Chemical Engineering, Shahrood Unit, Islamic Azad University, Shahrood, Iran
D. Norouzian
Department of Pilot Nano-Biotechnology, Pasteur Institute of Iran, Tehran-13164, Iran
B. Honarvar
Department of Chemical Engineering, Marvdasht Unit, Islamic Azad University, Marvdasht, Iran
M. Mohammadi
Department of Chemical Engineering, Marvdasht Unit, Islamic Azad University, Marvdasht, Iran
H. Ebrahimi Shamabadi
Department of Pilot Nano-Biotechnology, Pasteur Institute of Iran, Tehran-13164, Iran
A. Akbarzadeh
Department of Pilot Nano-Biotechnology, Pasteur Institute of Iran, Tehran-13164, Iran