ABSTRACT
Quantity of extracellular proteins and activities two cell wall degrading enzymes pectinase and cellulase were determined in the culture filtrate of Fusarium solani, the causal organism of root rot of Coleus forskohlii. Substitution of carbon source in the medium with either pectin or carboxymethyl cellulose led to the increased production of extracellular proteins by the fungus. Pectinase and cellulase activity in the culture filtrate was detected only when the growth medium contained substituted carbon source in the form of pectin and CMC, respectively. Pectinase activity was highest after 5 days incubation and then decreased gradually with time but cellulase activity showed a steady time dependent increase. In vitro virulence study showed the requirement of both the enzymes for complete expression of rot symptoms on Coleus plants. Thus the present study established the adaptive, substrate dependent expression of the two enzymes by the fungus and also their involvement in the root rot disease of Coleus forskohlii.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/pjbs.2013.2036.2040
URL: https://scialert.net/abstract/?doi=pjbs.2013.2036.2040
INTRODUCTION
Penetration of host surface is a critical step in the infection process of phytopathogenic fungi. Many plant pathogenic fungi penetrate into host cell by secreting large amount of extracellular enzymes such as pectinase, cellulase, xylanase and protease, which degrade plant cell wall (Tonukari, 2003; Agrios, 2005; Kikotet al., 2009). The expression of most cell wall degrading enzymes in vitro as well as in vivo depends on substrate availability (Tonukari, 2003; Hubballi et al., 2011). Coleus forskohlii Briq. is cultivated in several regions of India for harvesting its roots since it is the only source of forskolin, a medicinally important labdane diterpenoid (Bhat et al., 1977). The major problem observed in Coleus industry is due to the dreadful root rot disease causing severe damage in the quality and quantity of harvested raw material (Singh et al., 2011). To control any plant disease developing in a particular region, identification of the pathogen and understanding its interaction with the host is very important (Boland et al., 2004). In an earlier investigation, Bhattacharya and Bhattacharya, 2008 reported Fusarium solani (Mart.) Sacc. to be the causal organism of root rot of C. forskohlii from lower Gangetic West Bengal, India and also characterized the disease symptoms. Fusarium spp., secrete a number of hydrolytic enzymes capable of degrading cell wall polymers in order to invade the plant tissue (Roncero et al., 2003). Among different Plant Cell Wall Degrading Enzymes (PCDE), pectinases are important for the infection of various phytopathogenic fungi (Valette-Collet et al., 2003). Pectic enzymes are first induced when fungi are cultered on isolated plant cell walls and in infected tissue (Niture et al., 2006). Cellulases produced by several phytopathogenic fungi also play a role in the softening and disintegration of cell wall material (Agrios, 2005).
Enzyme production by phytopathogenic fungi is studied mostly on artificial growth media because of the presence of plant enzymes and microbial enzyme inhibitors that occur in the plants (Moreira et al., 2005). In vitro studies of fungal enzymes from culture filtrate have recently been used by several workers (Morid et al., 2009; Hubballi et al., 2011). Present study deals with the quantification of extracellular proteins and two cell wall degrading enzymes pectinase and cellulase from liquid submerged culture of Fusarium solani. Aim of the study was to understand the infection process of F. solani leading to the development of rot symptoms in C. forskohlii.
MATERIALS AND METHODS
Isolation of the fungal pathogen: Pathogenic F. solani was isolated from rot affected C. forskohlii plants and maintained on PDA by repeated sub-culture for further use in the study.
Preparation of culture filtrate: Culture filtrate of F. solani was prepared according to the method of Hubballi et al. (2011) in Czapek Dox Broth (CDB). The carbon source in the medium was substituted either with 1% pectin (for pectinase production) or 1% Carboxy Methyl Cellulose (CMC) (for cellulase production). Medium without pectin or CMC was used as control. The media (200 in 250 mL conical flasks) were inoculated with small mycelial discs (about 5 mm in diameter) from actively growing pure culture of F. solani. Five flasks were inoculated for each type of medium. Cultures were incubated at room temperature. Culture filtrates were obtained after 5, 10 and 15 days incubation. The broth was centrifuged at 3000 g for 20 min to remove mycelia and the cell free supernatant was used for further study.
Estimation of total soluble protein: Total soluble protein in the culture filtrates-Control (C), filtrate produced from pectin containing medium (F1) and from cellulose containing medium (F2), was determined by Lowry et al. (1951). Ten replicates were prepared for each sample including control and the experiment was repeated twice. Standard curve was prepared with Bovine Serum Albumin (BSA). Protein concentration in the unknown samples were calculated from linear portion of the standard curve and expressed as μg protein mL-1.
Determination of pectinase activity: Pectinase activity of C, F1 and F2 was determined by the standard protocol of Sigma (1997). All the reagents were prepared in double distilled water. Five percent pectin solution was used as substrate. The reagents were mixed in 50 mL Erlenmeyer flasks as follows:
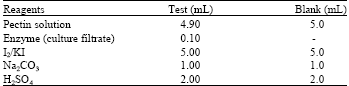 |
Before adding enzyme (culture filtrate), pectin solution was equilibrated to 25°C. After mixing enzyme the pectin-enzyme mixture was incubated at 25°C for exactly 5 min. After addition of the remaining reagents, mixtures were swirled. Then the blank and tests were titrated with reagent Na2S2O3 until the colour became light yellow. Finally a drop of starch indicator was added and continued titration with the same reagent until the solution became colourless. Volume of Na2S2O3 required for both blank and tests were recorded. The experiment was repeated five times. The enzyme activity was expressed as Units enzyme mL-1 culture filtrate and calculated from the formula:
Where:
| 1 | = | One μmole galacturonic acid is oxidized by 1 microequivalent of I2 |
| 100 | = | Microequivalents of S2O3 mL-1 of Na2S2O3. df = dilution factor, 5 = time of reaction in min |
| 0.1 | = | Volume of enzyme (culture filtrate) used, 2 = microequivalent of S2O3 oxidized per microequivalents of I2 reduced |
Determination of cellulase activity: Cellulase activity of C, F1 and F2 was determined spectrophotometrically by the method described by Sadasivam and Manickam (1996). The 0.45 mL of 1% CMC solution at a temperature of 55°C and 0.05 mL enzyme (culture filtrate) were mixed in test tube and incubated at 55°C in a water bath for 15 min. After removing the enzyme-substrate mixture from the bath, 0.5 mL 3, 5-Dinitrosalicylic Acid (DNS) reagent was added. Mixture was heated for 5 min in a boiling water bath. One Mililitter 40% potassium-sodium terterate was added to warm test tubes and cooled to room temperature. Volume was made up to 5 mL by adding distilled water. Absorbance was measured at 540 nm. A standard curve was prepared using glucose. Enzyme activity was expressed as μg glucose released/minute/μL culture filtrate.
Study of virulence of the culture filtrates: The virulence of culture filtrates was assessed on the basis of visible expression of rot symptoms on C. forskohlii shoots. Portions of shoots taken from in vitro raised healthy plants were treated in replicates of five with culture filtrates -C, F1, F2 and mixture of F1 and F2 (1:1) in sealed sterile petridish at room temperature. Shoots were also treated with sterilized CDB in a similar manner. Observations were made for appearance of disease symptoms after three days.
Statistical analysis: All statistical analyses were performed with Statistical Package for Social Sciences (SPSS) version 17 (SPSS Inc., Wacker Drive, Chicago, IL) (SPSS, 2007). Quantitative changes in different parameters were analyzed by analysis of variance (ANOVA) and mean separations were performed by post hoc analysis by Tucky’s HSD method.
RESULTS
Total soluble protein content: Total soluble protein content of culture filtrates F1 and F2 were significantly higher (at p = 0.05) than Control (C), irrespective of incubation periods (5, 10 and 15 days) (Table 1). Protein content in F1 and F2 after 10 days incubation increased significantly (approximately 4 times) than after 5 days.
| Table 1: | Total soluble protein content of culture filtrate of F. solani after different incubation periods |
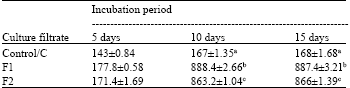 | |
| *Each value represents Mean±SE, **Means followed by same letters do not differ significantly at p = 0.05 ***Control: Culture filtrate from medium without pectin or CMC, F1: Culture filtrate from medium containing pectin, Culture filtrate from medium containing CMC | |
| Table 2: | Pectinase activity (units mL-1) of the culture filtrate of F. solani after different incubation periods |
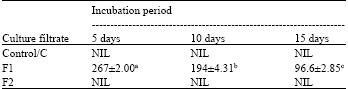 | |
| *Each value represents Mean ±SE, **Means followed by different letters differ significantly at p = 0.05, ***Control: Culture filtrate from medium without pectin or CMC, F1: Culture filtrate from medium containing pectin, Culture filtrate from medium containing CMC | |
But change was insignificant and almost negligible from 10 to 15 days. For C also, the pattern was similar. But increase from 5 days to 10 days was only 0.14 times. So presence of either of the substrates in the medium resulted in higher soluble protein content and also higher rate of increase in protein content of the culture filtrates, as compared to the control.
Pectinase and cellulase activity: Pectinase and cellulase activity was totally absent in control filtrate ‘C’ irrespective of incubation periods. But addition of only the appropriate substrate to the culture medium resulted in the induction of cellulase and pectinase activity. When pectin was the substrate, pectinase activity of F1 was highest after 5 days but then gradually reduced significantly with time (Table 2). Change in cellulase activity of F2, when CMC was the substrate, showed a reversed pattern and increased significantly with incubation period, lowest being after 5 days and highest after 15 days (Table 3).
Virulence of culture filtrates: Different types of culture filtrates showed variations in their ability to induce visible disease symptoms on C. forskohlii plants after 3 days incubation (Fig. 1). ‘CDB’ and ‘C’ were unable to induce any symptom at all (Fig. 1a and b, respectively). Expression of the disease was most severe on plants treated by a mixture (1:1) of F1 and F2 (Fig. 1e). F1 and F2 individually induced disease symptoms but to a lesser extent (Fig. 1c and d).
| Table 3: | Cellulase activity (μg glucose released/minute /μL culture filtrate) of the culture filtrate of F. solani after different incubation periods |
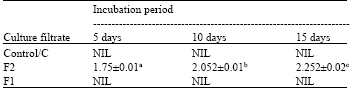 | |
| *Each value represents Mean ±SE, **Means followed by different letters differ significantly at p = 0.05, ***Control- Culture filtrate from medium without pectin or CMC, F1: Culture filtrate from medium containing pectin, Culture filtrate from medium containing CMC | |
 | |
| Fig. 1(a-e): | Effects of culture filtrates of F. solani on C. forskohlii, showing different degrees of disease expression (a) Plants treated with CDB showing no symptoms, (b) Plants treated with control culture filtrate (C) showing no symptoms (c) Partial rot symptoms developed in plants treated with F1, (d) Partial rot symptoms developed in plants treated with F2 and (e) Symptoms developed in plants treated with mixture (1:1) of F1+F2 |
DISCUSSION
Extracellular proteins secreted by fungus contain the enzymes corresponding to the types of glycosidic linkages present in the host cell wall polysaccharides to macerate tissues and degrade cell wall components (Moreira et al., 2005). All such cell wall splitting enzymes are secreted only in the presence of appropriate substrates (Hubballi et al., 2011). In the present study total soluble protein content of culture filtrate of F. solani was found to be significantly higher when the medium contained either of the two substrates, pectin or CMC and activities of two cell wall degrading enzymes (pectinase and cellulase) were detected in the culture filtrates of F. solani only when the growth medium contained appropriate substrates (Pectin and CMC, respectively). This was probably due to inductive effects of substrates. Hubballi et al. (2011) found the in vitro production of pectolytic and cellulolytic enzymes by Alternaria alternata only in the presence of pectin and cellulose, respectively in the medium. This corresponds with the finding of the present study indicating the adaptive secretion of cell wall degrading enzymes by F. solani in the presence of appropriate substrate.
According to Kikot et al. (2009), Fusarium sp. secrete a number of hydrolytic enzymes capable of degrading cell wall polymers in order to invade plant tissue. Morid et al. (2009) have the opinion that enzyme activity can be used as a predictive marker of pathogenicity and virulence of F. solani. Hubballi et al. (2011) are of opinion that the production and activity of pectinolytic and cellulolytic enzymes in vitro suggest their active role in disease development. Adaptive in vitro secretion of pectinase and cellulase by F. solani isolated from root rot infected C. forskohlii indicates the involvement of the enzymes in the disease.
In the present study pectinase activity was highest after 5 days incubation. Pectinase activity gradually reduced significantly after 10 and 15 days. Hubballi et al. (2011) also found a reduction in pectinase production in vitro by A. alternata after 10 days. Pectic enzymes are first polysaccharides to be induced when fungi are cultured on isolated plant cell wall or they infect plant tissue (Martinez et al., 1991; Niture et al., 2006). This is probably why pectinase activity was more in the initial stages and then decreased gradually. Cellulase activity of culture filtrate increased gradually with incubation period. Similar result was also obtained by Hubballi et al. (2011) for A. alternata. This suggests the increased involvement of cellulase in the advanced stages of the infection process.
Failure of control Culture filtrate (C) and CDB to induce any symptom at all was because of the absence of cell wall degrading hydrolytic enzymes pectinase and cellulase. Most severe expression of the disease on plants treated by a mixture (1:1) of F1 and F2 indicated that combined action of two types of enzymes was necessary for complete expression of disease symptoms.
The present study gives an idea about the mechanism of infection of F. solani on C. forskohlii and highlights the probable involvement of two cell wall degrading enzymes, pectinase and cellulase in the process. This is the first study of its kind with this particular host-pathogen combination that throws a light on the biochemistry of the disease development process.
REFERENCES
- Agrios, G.N., 2005. Plant Pathology. 5th Edn., Elsevier, Cambridge, Massachusetts, ISBN-13: 9780080473789, Pages: 922.
Direct Link - Boland, G.J., M.S. Melzer, A. Hopkin and V. Higgins, 2004. Climate change and plant diseases in Ontario. Canad. J. Plant Pathol., 26: 335-350.
CrossRefDirect Link - Hubballi, M., A. Sornakili, S. Nakkeeran, T. Anand and T. Raguchander, 2011. Virulence of Alternaria alternate Infecting noni associated with production of cell wall degrading enzymes. J. Plant Prot. Res., 51: 87-92.
CrossRefDirect Link - Kikot, E.G., A.R. Hours and M.T. Alconada, 2009. Contribution of cell wall degrading enzymes to pathogenesis of Fusarium graminearumi: A review. J. Basic Microbiol., 49: 231-241.
CrossRefPubMedDirect Link - Lowry, O.H., N.J. Rosebrough, A.L. Farr and R.J. Randall, 1951. Protein measurement with the folin phenol reagent. J. Biol. Chem., 193: 265-275.
CrossRefPubMedDirect Link - Martinez, M.J., T.M. Alconada, F. Guillen, C. Vazquez and F. Reyes, 1991. Pectic activities from Fusarium oxysporum f. sp. melonis. Purification and characterization of an exopolygalacturonase. FEMS Microbial. Lett., 81: 145-150.
Direct Link - Moreira, F.G., S. dos Reis, M.A.F. Costa, C.G.M. de Souza and R.M. Peralta, 2005. Production of hydrolytic enzymes by the plant pathogenic fungus Myrothecium verrucaria in submerged cultures. Brazil. J. Microbiol., 36: 7-11.
CrossRefDirect Link - Morid, B., R. Zare, S. Rezaee, H. Zamani-Zadeh and S. Hajmansour, 2009. The relationship between cutinases and the pathogenicity/virulence of Fusarium solani in potato tubers. Phytopathol. Mediterr., 48: 403-410.
Direct Link - Niture, S.K., A.R. Kumar and A. Pant, 2006. Role of glucose in production and repression of polygalacturonase and pectate lyase from phytopathogenic fungus Fusarium moniliforme NCIM 1276. World J. Microb. Biotech., 22: 893-899.
CrossRefDirect Link - Singh, R., S.P. Gangwar, D. Singh, R. Singh, R. Pandey and A. Kalra, 2011. Medicinal plant Coleus forskohlii Briq : Disease and management. Med. Plants, 3: 1-7.
Direct Link - Roncero, M.I.G., C. Hera, M. Ruiz-Rubio, F.I.G. Maceira and M.P. Madrid et al., 2003. Fusarium as a model for studying virulence in soilborne plant pathogens. Physiol. Mol. Plant Pathol., 62: 87-98.
CrossRef - Tonukari, N.J., 2003. Enzymes and fungal virulence. J. Applied Sci. and Environ. Manage., 7: 5-8.
Direct Link








