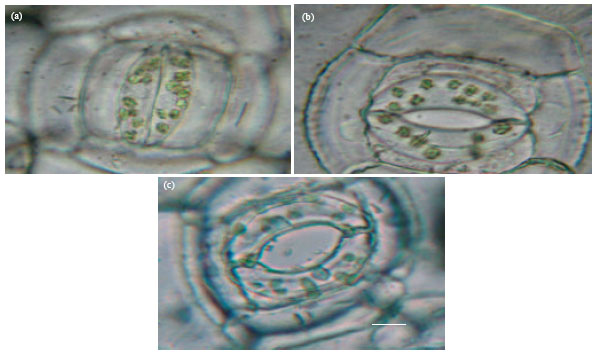
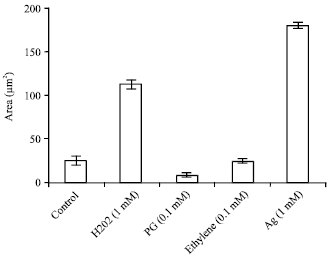
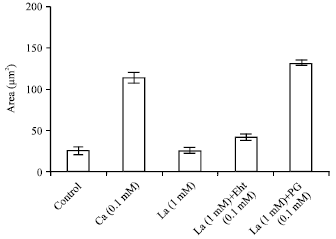
Research Article
Differential Role of Ethylene and Hydrogen Peroxide in Dark-induced Stomatal Closure
Plant Physiology and Biochemistry Laboratory, Department of Botany, Visva-Bharati University, Santiniketan-731 235, West Bengal, India
N. Parvin
Department of Botany, Biology-1, Biocentre, Ludwig Maximillians, University of Munich, 82152 Martinsried, Germany
D. Laha
Centre for Plant Molecular Biology, Eberhard Karls, University of Tuebingen, Auf der Morgenstelle 1, 72076 Tuebingen, Germany