ABSTRACT
In the present study, seasonal variation of vitamin and sterol content of Chironomidae larvae were determined by using HPLC. As the result of vitamin analysis, we found α-tocopherol, retinol, K1, K2, D2 and D3. When the seasonal variation of vitamin groups were compared, a significant increase was observed in vitamin K1, K2, D2 and α-tocopherol in all seasons. A significant increase was observed in vitamin D3 in spring. And also vitamin A level high in autumn and winter. α-tocopherol level was significantly high among vitamins. When vitamin groups were compared statistically, differences were detected between seasons (p<0.001). Analyzing the content of sterol, we found ergosterol, cholesterol, stigmasterol and β-sitosterol in all seasons. Cholesterol level was found to be significantly high in sterols. When sterol contents were compared statistically, differences were detected between seasons (p<0.001). In conclusion, the reasons for these differences are larval development feature and the variety of food in different seasons.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/pjbs.2013.1579.1583
URL: https://scialert.net/abstract/?doi=pjbs.2013.1579.1583
INTRODUCTION
Insects play an important role in food chain of aquatic system and among Diptera the Chironomid larvae (midge larvae) are recognized as an important food item for many fishes and cultured invertebrate (Shaw and Mark, 1980; Habib et al., 1997; Yusoff et al., 1996; Fernando, 1994; Tidwell et al., 1997; Wolfram-Wais et al., 1999). Chironomid larve are excellent source of lipid, vitamins and minerals (McLarney et al., 1974).
Field observation with some Chironomid larvae showed that environmental factors such as temperature, pH, toxic substances, photoperiod, oxygen content and biotic interaction may influence growth in the Chironomidae (Tokeshi, 1995). Benthic communities of the profundal regions of freshwater ecosystems are dependent on sedimentation processes for their food supply (Brinkhurst, 1974). Food availability and feeding biology are two important factors determining seasonal changes in food ingestion, growth and reproduction of Chironomid larvae (Jonasson, 1972). Several studies have shown that Chironomus growth is closely correlated with the availability of algae or detritus of algal origin in their food (Kajak and Warda, 1968; Jonasson, 1972; Lindegaard and Jonasson, 1979; Johnson and Pejler, 1987).
Insects, protein, mineral, vitamin and carbohydrates need (Hagen et al., 1974). Sterols are required for cell membrane and are produced by plants and animals. B-sitosterol, stigmasterol and ergosterol are abundant in plants. Cholesterol, kaprosterol and allosterol are sterols in the oils of animal origin (Liu, 2003). ADEK vitamins are fat-soluble vitamins. Vitamin E functions as an antioxidant in cell membrane and lipoproteins (Keskin, 1987; Belitz et al., 2005).
The aim of this study is to analyze of vitamin and sterol content of Chironomidae larvae and then statistically compare the variation between seasons.
MATERIALS AND METHODS
Chironomid larvae were collected on spring, summer, autumn and winter 2010 in Büyük stream (Pelte/Elazig). Then samples were stored at deep-freeze until analysis. The weights of samples were weighed and then were put into tubes. Vitamins and sterols were extracted from lipid extract by the method of Sanchez-Machado et al. (2004) and Lopez-Cervantes et al. (2006) with minor modifications. Five mL n-hexane/isopropyl alcohol mixture was treated 5 mL KOH solution (0.5 M in methanol) were added and immediately vortexed for 20 sec. The tubes were placed in a water bath at 80°C for 15 min. Then after cooling in iced water, 1 mL of distilled water and 5 mL of hexane was added and the mixture was rapidly vortexed for 1 min, then centrifuged for 5 min at 5000 rpm. The supernatant phase were transferred to another test tube and dried under nitrogen. The residue was redissolved in 1 mL of the HPLC mobile phase (68:24:4 (v/v/v) methanol: acetonitrile: water). Finally, an aliquot of 20 μL was injected into the HPLV column. Before injection, the extracts were maintained at -20°C away from light.
Chromatographic analysis was performed using an analytical scale (15x0.45 cm I.D) Supelco LC 18TM column with a particle size 5 μm (sigma, USA). HPLC conditions were as follows: mobile phase 60:38:2 (v/v/v): acetonitrile/methanol/water; a flow rate of 1 mL min-1; column temperature 30°C. The detection was operated using two channels of a diode-array spectrophotometer, 326 nm for retinol, 265 nm for vitamin D and vitamin K, 202 nm for alpha-tocopherol and phytosterols (Lopez-Cervantes et al., 2006).
Statistical analysis was performed using SPSS software (ver. 10.0). The experimental results were reported as Mean±SEM (standard error of means). Analysis of variance (ANOVA) and an LSD (least significant difference) test were used to compare the experimental groups.
RESULTS
When the vitamin groups were compared, α-tocopherol level was found to be significantly high among vitamins. When α-tocopherol was compared between seasons, a significant increase was observed on summer (p<0.001) (Table 1).
Partial increases were observed in vitamin K1 and vitamin D3 in spring (p<0.001). And also partial increases was observed in K2 in summer (p<0.05). When the seasonal variation of vitamin content were compared, a significant increase were observed in vitamin retinol and vitamin D2 in summer (p<0.01, p<0.05). On the other hand, vitamin content was decreased in autumn when vitamin groups compared statistically between seasons (p<0.001, p<0.01, p<0.05) (Fig. 1).
When compared of sterol contents of Chironomidae larvae, were found a significant increase in cholesterol and stigmasterol level (p<0.001). Cholesterol, stigmasterol and β-sitosterol increased significantly in summer according to other seasons. Cholesterol level was found to be significantly high among sterols. Unlike partially increase was observed in ergosterol in winter (p<0.05).
DISCUSSION
When the vitamin groups were compared, α-tocopherol level was found to be significantly high among vitamins and in summer (p<0.001). And when sterol groups were compared statistically, cholesterol level was found to be significantly high in all seasons and especially in summer (p<0.001) (Fig. 2). We think feeding activity and development properties of Chironomidae larvae as the cause of this situation. Especially during summer months, larvae feed on plant origin food.
| Table 1: | Seasonal variations of vitamin and sterol content of chironomidae larvae (mg g-1) |
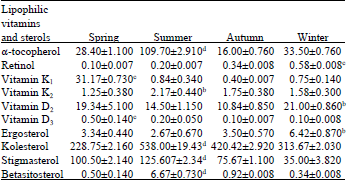 | |
| a: p>0.05, b: p<0.05, c: p<0.01, d: p<0.001 | |
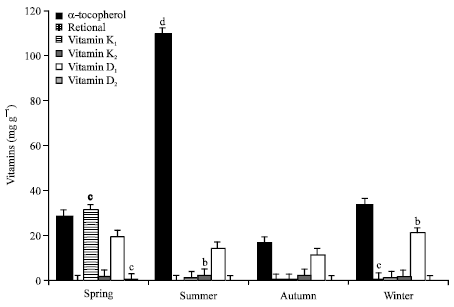 | |
| Fig. 1: | Levels of vitamins in chironomidae larvae (mg g-1), a: p>0.05, b: p<0.05, c: p<0.01, d: p<0.001 |
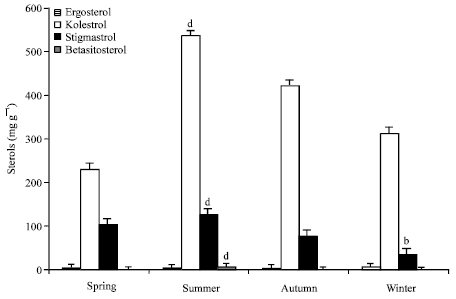 | |
| Fig. 2: | Levels of sterols in chironomidae larvae (mg g-1), a: p>0.05, b: p<0.05, c: p<0.01, d: p<0.001 |
It is known that insects contain almost entirely cholesterol, even if their food contains significant amounts of phytosterols. This is due to dealkylation of dietary phytosterols to cholesterol, a process, characteristic for Arthropoda (Behmer and Elias, 2000). Stefanov et al. (2002) analyzed lipid and sterols of Musca domestica L. larvae. They obtained the opposite results cholesterol was comparatively low for insects and phytosterols appeared in significant concentrations, suggesting that phytosterols are dealkylated slowly in Musca domestica larvae. In contrast to we determined cholesterol level was high Chironomidae larvae (Table 1).
Also Stefanov et al. (2002) checked the possibility that phytosterols are derived from the diet, so they investigated the sterol composition of dry milk dissolved in water which is the food of larvae. They found very low sterol concentrations, with cholesterol accounting for more than 90% of the total sterol mixture. Less than 3-4 % sitosterol and campesterol were present. Under these conditions the only alternative source for phytosterols is the wheat bran. Observations of the midgut contents showed the presence of chewings, originating from the wheat bran. It is possible that the larvae are able to scrape the substrate with their mouthparts (Sareen et al., 1990). These findings, difference feeding Chironomidae larvae, is evidence that showing how it affect of vitamin and sterol content of larvae.
Vitamin E was included in the diet of some insects for its antioxidant activities to protect the integrity of fatty acids and perhaps of other substances (Fraenkel and Blewett, 1946; Beck et al., 1949; Vanderzant, 1957). Fraenkel and Blewett (1946) found that vitamin E improved the growth of Anagasta (Ephestia) kühniella (Zeller), but they reasonably favoured the supposition that its role was principally as a protective antioxidant on unsaturated fatty acids rather than as a nutritional requirement of the insect.
Both vitamin A and vitamin E increased the rate of larval growth and improved the rate of development of pupae so as to increase the number of adult emergent (House, 1951; Coppel et al., 1959). House (1966) studied effects of vitamin E and vitamin A on growth and development Agria affinis larvae (Diptera) and determined vitamin E is requirement for reproduction and vitamin A increased growth. Vitamin E (α-tocopherol) level was found significantly high in our study (Fig. 1). As the cause of this situation, is shown Chironomidae larvae stored vitamin E for later stages of development.
Several studies have demonstrated the importance of several pulses of pelagic algal detritus to Chironomus growth (Hilsenhoff, 1966; Jonasson and Kristiansen, 1967; Jonasson, 1972; Kajak, 1977; Lindegaard and Jonasson, 1979; Johannsson, 1980; Johnson and Pejler, 1987). Furthermore, invertebrate growth has been shown to be slower when detritus is the dominating dietary constituent, whereas the ingestion of algae has been positively correlated with increased growth (Cummins, 1973). One study was showed that blue-green algae are a major portion (68% of gut contents) of the diet of filter feeding Chironomus crassicaudatus (Ali, 1990). Benthic green algae have been reported to be a major food source as stated by Brook (1954) who observed that Chironomidae larvae on a sand-filter bed ingested only filamentous blue green algae and filamentous diatoms.
In conclusion, we determined seasonal variation of vitamin and sterol content of Chironomidae larvae. We have detected difference in vitamin and sterol content between seasons. As the cause of this situation, we think the reasons for these differences are larval development feature and the variety of food in different seasons. Our findings and our research is new for our country. Therefore, these findings are important for biochemical analysis of future.
ACKNOWLEDGMENTS
In this study, vitamin and sterol analysis was performed in biochemistry laboratory. We thank for Okkes Yilmaz for his assistance in this study.
REFERENCES
- Ali, A., 1990. Seasonal changes of larval food and feeding of Chironomus crassicaudatus (Diptera: Chironomidae) in a subtropical lake. J. Am. Mosquito Control Assoc., 6: 84-88.
Direct Link - Beck, S.D., J.H. Lilly and J.F. Stauffer, 1949. Nutrition of the European corn borer, Pyrausta nubialis (Hbn.) I. development of a satisfactory purified diet for larval growth. Ann. Entomol. Soc. Am., 42: 483-496.
Direct Link - Behmer, S.T. and D.O. Elias, 2000. Sterol metabolic constraints as a factor contributing to the maintenance of diet mixing in grasshoppers (Orthoptera: Acrididae). Physiol. Biochem. Zool., 73: 219-230.
PubMedDirect Link - Brook, A.J., 1954. The bottom-living algal flora of slow sand filter beds of waterworks. Hydrobiologia, 6: 333-351.
CrossRef - Coppel, H.C., H.L. House and M.G. Maw, 1959. Studies on dipterous parasites of the spruce budworm, Choristoneura fumiferana (Clem.) (Lepidoptera: Tortricidae): VII. Agria affinis (Fall.) (Diptera : Sarcophagidae). Can. J. Zool., 37: 817-830.
CrossRefDirect Link - Cummins, K.W., 1973. Trophic relations of aquatic insects. Annu. Rev. Entomol., 18: 183-206.
CrossRefDirect Link - Fernando, C.H., 1994. Zooplankton, fish and fisheries in tropical freshwaters. Hydrobiologia, 272: 105-123.
CrossRef - Fraenkel, G. and M. Blewett, 1946. Linoleic acid, vitamin E and other fat-soluble substances in the nutrition of certain insects, Ephestia kuehniella, E. elutella, E. cautella and Plodia interpunctella (Lep.). J. Exp. Biol., 22: 172-190.
Direct Link - Habib, M.A.B., F.M. Yusoff, S.M. Phang, K.J. Ang and S. Mohamed, 1997. Nutritional values of chironomid larvae grown in palm oil mill effluent and algal culture. Aquaculture, 158: 95-105.
CrossRefDirect Link - Hilsenhoff, W.L., 1966. The biology of Chironomus plumosus (Diptera: Chironomidae) in lake Winnebago, Wisconsin. Ann. Entomol. Soc. Am., 59: 465-473.
Direct Link - House, H.L., 1966. Effects of vitamins E and A on growth and development and the necessity of vitamin E for reproduction in the parasitoid Agria affinis (Fallen) (Diptera: Sarcophagidae). J. Insect Physiol., 12: 409-417.
CrossRefDirect Link - Johannsson, O.E., 1980. Energy dynamics of the eutrophic chironomid Chironomus plumosus f. semireductus from the Bay of Quinte, Lake Ontario. Can. J. Fish. Aquat. Sci., 37: 1254-1265.
CrossRefDirect Link - Jonasson, P.M. and J. Kristiansen, 1967. Primary and secondary production in Lake Esrom. Growth of Chironomus anthracinus in relation to seasonal cycles of phytoplankton and dissolved oxygen. Int. Rev. Gesamten Hydrobiol. Hydrogr., 52: 163-217.
CrossRefDirect Link - Kajak, Z., 1977. Factors influencing benthos biomass in shallow lake environments. Ekol. Polska, 25: 421-429.
Direct Link - Lindegaard, C. and P.M. Jonasson, 1979. Abundance, population dynamics and production of zoobenthos in Lake Myvatn, Iceland. Oikos, 32: 202-227.
Direct Link - Liu, R.H., 2003. Health benefits of fruit and vegetables are from additive and synergistic combinations of phytochemicals. Am. J. Clin. Nutr., 78: 517S-520S.
CrossRefPubMedDirect Link - Lopez-Cervantes, J., D.I. Sanchez-Machado and N.J. Rios-Vazquez, 2006. High-performance liquid chromatography method for the simultaneous quantification of retinol, α-tocopherol, and cholesterol in shrimp waste hydrolysate. J. Chromatogr. A, 1105: 135-139.
CrossRefPubMedDirect Link - McLarney, W.O., S. Henderson and M.M. Sherman, 1974. A new method for culturing Chironomus tentans Fabricius larvae using burlap substrate in fertilized pools. Aquaculture, 4: 267-276.
CrossRefDirect Link - Sareen, M.L., T.P. Kaur and S.P. Kaur, 1990. Some biochemical aspects of the postembriyonic development of Musca domestica L. (Diptera: Muscidae). Environ. Ecol., 8: 339-343.
Direct Link - Shaw, P.C. and K.K. Mark, 1980. Chironomid farming-a means of recycling farm manure and potentially reducing water pollution in Hong Kong. Aquaculture, 21: 155-163.
CrossRef - Stefanov, K., J. Nechev, G. Lavchieva-Nacheva, N. Nikolova and K. Seizova et al., 2002. Lipids and sterols in Musca domestica L. (Diptera, Muscidae): Changes after treatment with sucrose and lead. Comp. Biochem. Physiol. Part B: Biochem. Mol. Biol., 131: 543-550.
CrossRefDirect Link - Sanchez-Machado, D.I., J. Lopez-Hernandez, P. Paseiro-Losada and J. Lopez-Cervantes, 2004. An HPLC method for the quantification of sterols in edible seaweeds. Biomed. Chromatogr., 18: 183-190.
CrossRefPubMedDirect Link - Tidwell, J.H., G. Schulmeister, C. Mahl and S. Coyle, 1997. Growth, survival, and biochemical composition of freshwater prawns Macrobrachium rosenbergii fed natural food organisms under controlled conditions. J. World Aquacult. Soc., 28: 123-132.
CrossRef - Wolfram-Wais, A., G. Wolfram, B. Auer, E. Mikschi and A. Hain, 1999. Feeding habits of two introduced fish species (Lepomis gibbosus, Pseudorasbora parva) in neusiedler see (Austria), with special reference to chironomid larvae (Diptera: Chironomidae). Hydrobiologia, 408: 123-129.
CrossRef








