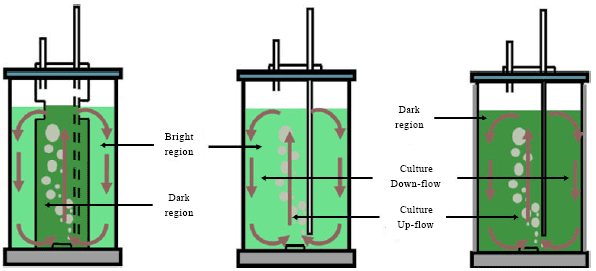
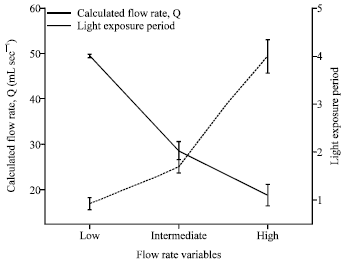
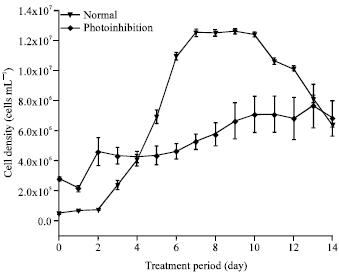
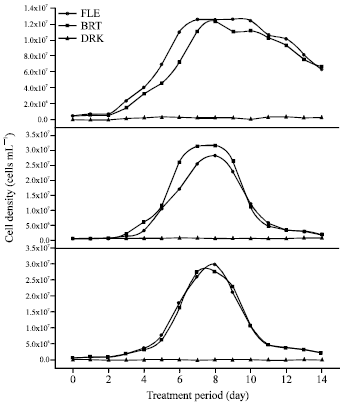
Research Article
Flashing Light as Growth Stimulant in Cultivation of Green Microalgae, Chlorella sp. Utilizing Airlift Photobioreactor
Department of Engineering Science, Faculty of Science and Technology, Universiti Malaysia Terengganu, Jln. Tok Jembal, 21030, Terengganu, Malaysia
A.H. Siti Hajar
Department of Engineering Science, Faculty of Science and Technology, Universiti Malaysia Terengganu, Jln. Tok Jembal, 21030, Terengganu, Malaysia
D. Wan Nur Sakinah
Department of Engineering Science, Faculty of Science and Technology, Universiti Malaysia Terengganu, Jln. Tok Jembal, 21030, Terengganu, Malaysia
Z. Nurhazwani
Department of Engineering Science, Faculty of Science and Technology, Universiti Malaysia Terengganu, Jln. Tok Jembal, 21030, Terengganu, Malaysia
J. Ahmad
Institute of Tropical Aquaculture (AKUATROP), Universiti Malaysia Terengganu, Jln. Tok Jembal, 21030, Terengganu, Malaysia