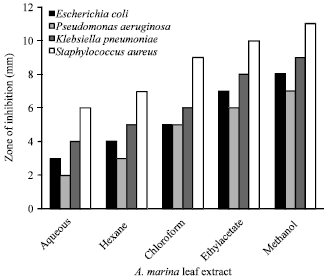
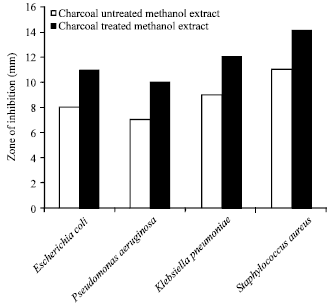
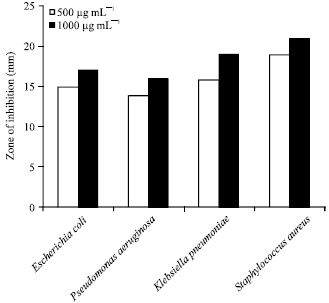
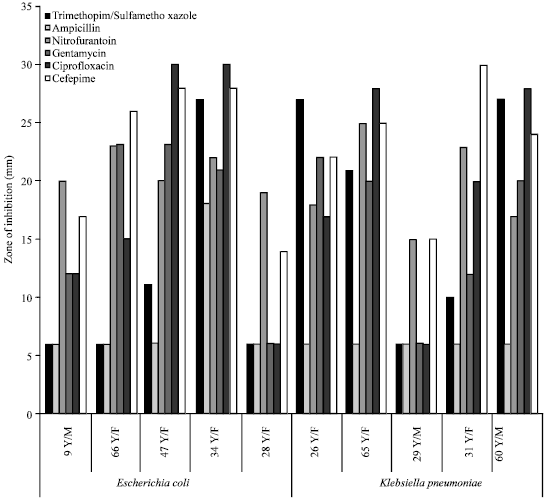
Research Article
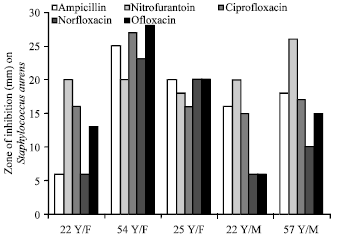
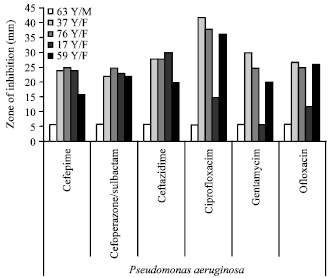
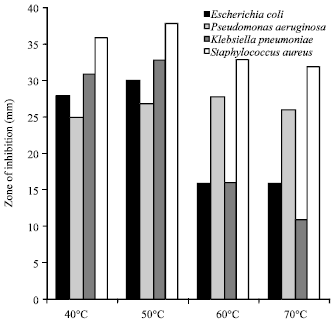
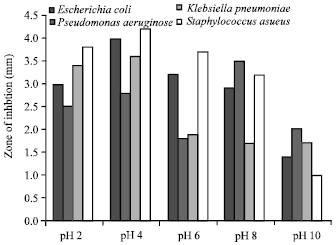
In vitro Antibacterial Activity and Stability of Avicennia marina against Urinary Tract Infection Pathogens at Different Parameters
Department of Biotechnology, Karpaga Vinayaga College of Engineering and Technology, Chinna Kolambakkam, Palayanoor Post, Madurantagam Taluk, Kanchipuram, 603308, Tamilnadu, India
Johanna Rajkumar
Department of Biotechnology, Rajalakshmi Engineering College, Thandalam, Chennai, Tamilnadu, India