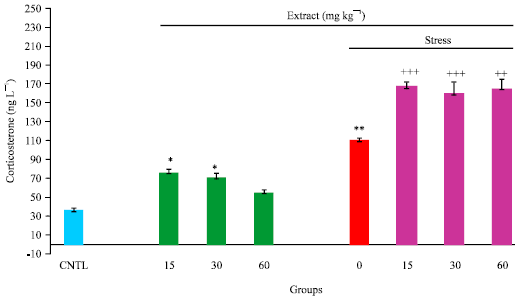
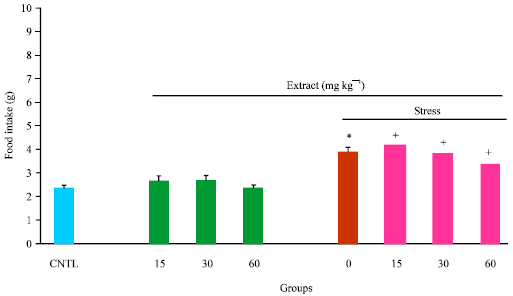
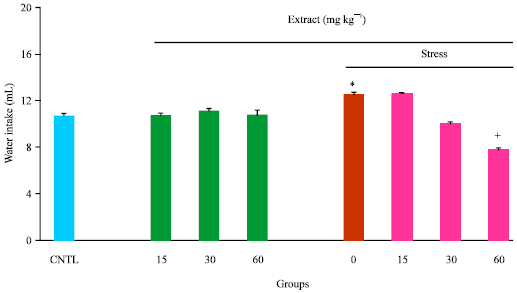
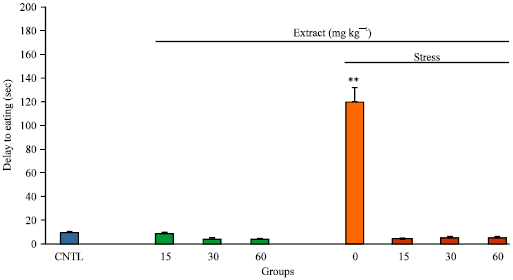
Research Article
Reduction of Metabolic Signs of Acute Stress in Male Mice by Papaver rhoaes Hydro-alcoholic Extract
Neurociences Research Center, Baqiyatallah (A.S.) University of Medical Sciences, Tehran, Iran
P. Mirzaei
Department of Psychology, School of Psychology, Roodhen Branch, Islamic Azad University, Tehran, Iran
F. Lotfi
Department of Psychology, School of Psychology, Roodhen Branch, Islamic Azad University, Tehran, Iran
S. Behzadi
Department of Psychology, School of Psychology, Roodhen Branch, Islamic Azad University, Tehran, Iran
H. Sahraei
Neurociences Research Center, Baqiyatallah (A.S.) University of Medical Sciences, Tehran, Iran