Research Article
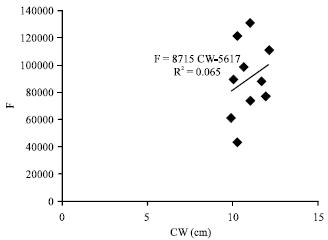
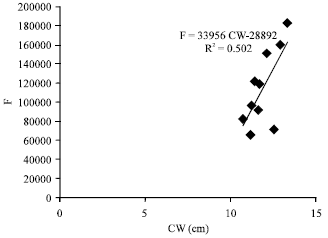
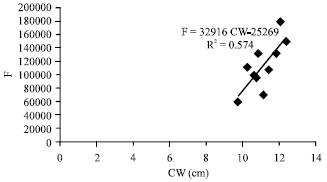
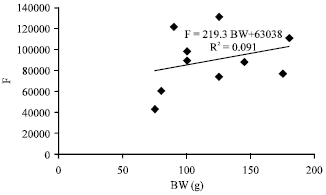
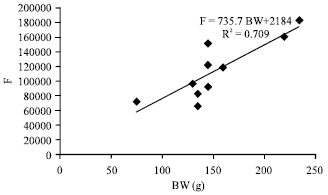
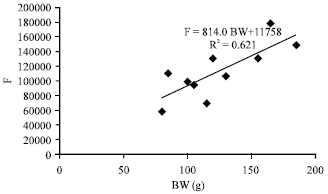
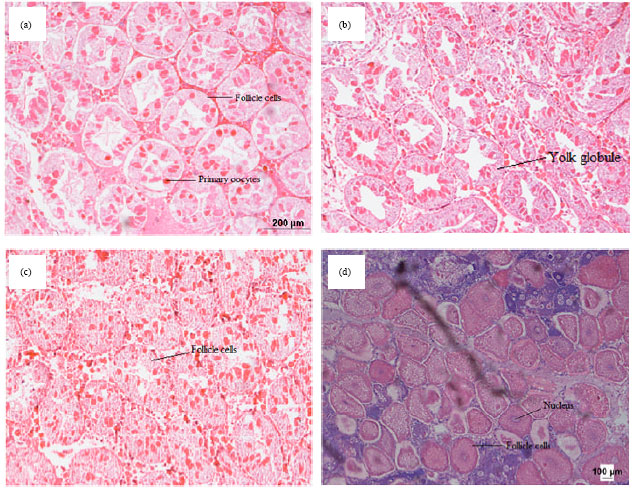
Fecundity, Embryonic and Ovarian Development of Blue Swimming Crab, Portunus pelagicus (Linnaeus, 1758) in Coastal Water of Johor, Malaysia
Institute of Tropical Aquaculture, Universiti Malaysia Terengganu, Kuala Terengganu, Malaysia
M.N. Azra
Institute of Tropical Aquaculture, Universiti Malaysia Terengganu, Kuala Terengganu, Malaysia
H. Siti-Aimuni
Faculty of Fisheries and Aqua-Industries, Universiti Malaysia Terengganu, Kuala Terengganu, Malaysia
A.B. Abol-Munafi
Faculty of Fisheries and Aqua-Industries, Universiti Malaysia Terengganu, Kuala Terengganu, Malaysia