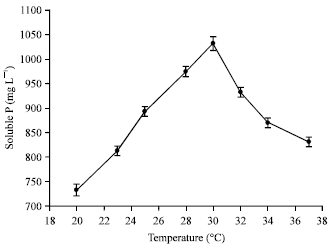
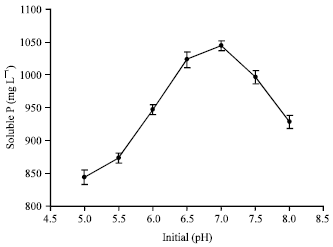
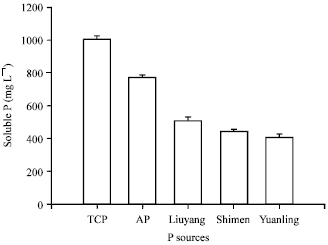
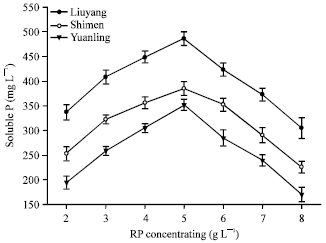
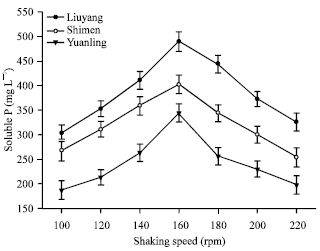
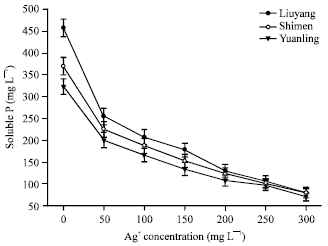
Short Communication
Isolation of Phosphate-solubilizing Fungus and Its Application in Solubilization of Rock Phosphates
Department of Environmental Microbiology, Hunan Institute of Microbiology, Changsha, Hunan 410009, China
He Yuelin
Department of Environmental Microbiology, Hunan Institute of Microbiology, Changsha, Hunan 410009, China
Yin Hongmei
Department of Environmental Microbiology, Hunan Institute of Microbiology, Changsha, Hunan 410009, China
Chen Wei
Department of Environmental Microbiology, Hunan Institute of Microbiology, Changsha, Hunan 410009, China
Wang Zhen
Department of Environmental Microbiology, Hunan Institute of Microbiology, Changsha, Hunan 410009, China
Xu Lijuan
Department of Environmental Microbiology, Hunan Institute of Microbiology, Changsha, Hunan 410009, China
Zhang Aiqun
Department of Environmental Microbiology, Hunan Institute of Microbiology, Changsha, Hunan 410009, China