ABSTRACT
Microalgae exhibit a number of heavy metal uptake process by different metabolism. In this study, the ability of microalgae for removal of heavy metal from wastewater was studied. Growth and biochemical contents of microalgae were determined by spectrophotometer. Heavy metal analysis of wastewater effluents were performed by atomic absorption spectrophotometer before and after treatment at laboratory scale. The growth of Scenedesmus bijuga and Oscillatoria quadripunctulata in sewage wastewater was higher than those grown in synthetic medium. Whereas, the growth of S. bijuga and O. quadripunctulata in sterilized petrochemical effluents was slightly lower than that grown in the standard synthetic medium. The chlorophyll, carotenoid and protein content of S. bijuga and O. quadripunctulata grown in sterilized sewage wastewater were higher than those grown in the standard medium. Similarly S. bijuga and O. quadripunctulata grown in sterilized petrochemical effluents showed lower contents of pigments and protein than those grown in sewage and synthetic medium. Heavy metals copper, cobalt, lead and zinc were removed by 37-50, 20.3-33.3, 34.6-100 and 32.1-100%, respectively from sewage wastewater and petrochemical effluent using Ocillatoria culture. The metal absorption by S. bijuga were (Cu, Co, Pb, Zn) 60-50, 29.6-66, 15.4-25 and 42.9-50%, respectively from sewage and petrochemical effluents. Both species showed high level of heavy metal removal efficiency and metal sorption efficiency of both microalgae depended on the type of biosorbent, the physiological status of the cells, availability of heavy metal, concentration of heavy metal and chemical composition of wastewater.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/pjbs.2011.805.811
URL: https://scialert.net/abstract/?doi=pjbs.2011.805.811
INTRODUCTION
Untreated domestic and industrial wastewater contaminates freshwater bodies by releasing huge amounts of wastewater with high BOD and COD loading whereas, industrial effluents add various heavy metals (Cd, Cr, Co, Cu, Pb, Mn, Zn) in it, resulting in high levels of water pollution. Most of the Indian rivers and freshwater streams are seriously polluted by industrial wastewater which come out of different factories. Although volume wise, the domestic sewage constitutes about 75% of the total effluent generated, it is the industrial effluent which contains high concentrations of pollutants, either toxic or non-toxic, that is of greater concern. Amount of water used in industry turns into wastewater polluting the surface and ground water and creating health hazards. If 50% (495 billion m3) of this consumed water is available for algae production, it could generate ~247 million tons of algal biomass and 37 million tons of oil. They also can supply O2 to heterotrophic aerobic bacteria to mineralize pollutants, such as hydrocarbons of petroleum (Huang et al., 2005; He and Xue, 2010), toxic by-products of the industry (Wang et al., 2006) and heavy metals such as Cr, Cu, Fe, Mn, Ni and Zn (Haritonidis and Malea, 1999) from the environment or for rendering them to harmless species is defined as phytoremediation (Pilon-Smits, 2005).
Fast growing petrochemical refineries are the source of toxic effluents entering the watercourses throughout the world. The products of industry are categorized as aliphatic, cyclic aliphatic, aromatic and inorganic compounds. As not all refineries have the same processes, the effluents that are produced will have different chemical compositions depending on the type of treatment they receive (Han et al., 2007). Petroleum refinery wastewaters are made up of many different chemicals which include oil and greases, phenols (creosols and xylenols), sulphides, ammonia, suspended solids, cyanides, nitrogen compounds and heavy metals like chromium, iron, nickel, copper, molybdenum, selenium, vanadium and zinc (Lehtinen, 1986).
Many species of microalgae are able to effectively grow in wastewater conditions through their ability to utilize abundant organic carbon and inorganic N and P in the wastewater. This solar driven technology is low-cost when compared to other available physical and chemical remediation methods (Cote, 1976).
The efficient growth of microalgae in wastewater depends on a variety of variables. As with any growth medium, critical variables are the pH and temperature of the growth medium, the concentration of essential nutrients, including N, P and organic carbon (and the ratios of these constituents) and the availability of light, O2 and CO2. Chlorella pyrenoidosa had a high protein content when grown on sewage sludge (Azmat et al., 2005) and the aqueous extract retained a comparatively low level of various heavy metals, i.e. Cu2+, Mn2+, Fe2+ and Zn2+ (Wong and Tam, 1984; Khorramabadi and Cheshmeh Soltani, 2008). Marine algae treated sludge can be used for vegetable yield of Allium cepa and Spinacia oleraceae was reported by Azmat et al. (2007). This research aimed to study the ability of microalgae for removal of pollutants from sewage and petrochemical industrial effluents for eventual goal to recycling water.
MATERIALS AND METHODS
Wastewater sampling: The sewage wastewater was collected from industrial city located at Noyyal river, Tirupur. The petrochemical effluent was collected from effluent treatment system of the oil refinery of Kochi, Kerala state during the month of May, 2011. The source of petrochemical effluent was collected from trickling filtered fire pond.
Microorganisms used: The test organisms selected for present investigation, Scenedesmus bijuga and Oscillatoria quadripunctulata were isolated from the petrochemical fire pond discharge river catchment area and the organisms were identified according to Prescott (1978).
Culture medium and conditions: The S. bijuga in Ward and parish medium (Ward and Parrish, 1982) and O. quadripunctulata in BG11 medium (Stanier et al., 1971) were maintained. The culture was incubated at 28-30°C under continuous light for 15 days. The cultures were aerated with dry sterilized air which contained about 3% CO2. Two species of algae were exposed to both effluents for 15 days. As a control Ward-Parish medium and BG11 medium were used for S. bijuga and O. quadripunctulata cultivation, respectively.
Biological analysis
Growth analysis: The growth was assessed spectrophotometrically at 750 nm (Novaspec LKB). The microalgal growth was also determined by the dry weight. A definite volume of algal suspension, after being centrifuged, dried overnight in an oven at 105°C. The data were given as mg L-1 of algal suspension.
Estimation of pigments (chlorophyll a and carotenoid): Chlorophyll content was determined spectrophotometrically, following extraction of the cells using 80% methanol. Chlorophyll a was determined in the methanol extracts using the equation by MacKinney (1941).
Estimation of proteins: Protein content concentration was determined by the method of Bradford (1976) protein was colorimetrically measured at 760 nm against blank using spectrophotometer and calibrated with bovine serum albumin as a standard.
Chemical analysis: The water samples were analyzed for pH using a pH meter (Elico 120 I). Heavy metals like Copper (Cu), Cobalt (Co), Lead (Pb) and Zinc (Zn) were determined as per standard methods (APHA, 1998). A Unicam 919 model atomic absorption spectrophotometer was also used for the determination of the heavy metals.
Statistical analysis: One-way analyses of variance (p<0.05) were used according to SPSS 16.0 version, 2011.
RESULTS
Effect of sewage wastewater on the growth of S. bijuga and O. quadripunctulata: Figure 1 shows that the dry weight of the S. bijuga grown in both sewage wastewater and Ward-Parish medium increased with time up to 12 days, then became down till the end of incubation period but wastewater seemed to support growth better than the tested medium. The highest biomass obtained at the 12th days of incubation was 320 mg L-1 of the cultures grown in wastewater corresponding to 260 mg L-1 in Ward and Parish medium. The dry weight of the organism increased by 23.07% in cultures grown on wastewater as compared to the standard medium.
The results in Fig. 2 indicates that there was a considerable amount of biomass of O. quadripunctulata grown in wastewater reached 441 mg L-1 at the 12th day of incubation period corresponding to 310 mg L-1 in BG11 medium. The dry weight of the organism increased by 42.25% in cultures grown on wastewater as compared to the standard medium.
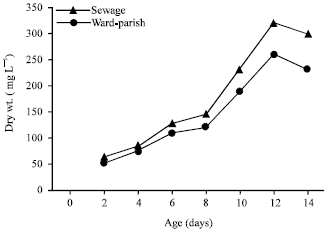 | |
| Fig. 1: | Growth of S. bijuga on sewage wastewater and Ward-Parish medium |
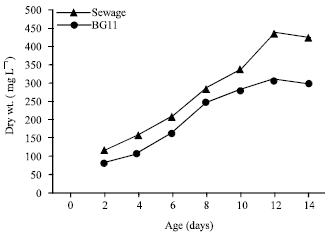 | |
| Fig. 2: | Growth of O. quadripunctulata on sewage wastewater and BG11 medium |
Effect of petrochemical effluent on the growth of S. bijuga and O. quadripunctulata: Figure 3 shows that the mean dry weight of S. bijuga in both standard medium and effluent water recorded more or less the same growth pattern. The highest biomass obtained at 12 days of incubation was 322 mg L-1 in standard medium corresponding to 315 mg L-1 in petrochemical effluent. Therefore, effluent water reduced the growth by 2.2% compared to Standard medium.
Data in Fig. 4 show that the growth of O. quadripunctulata increased in both the petrochemical effluent and the standard medium. The trend of increase continued up to the 10th days of incubation. At the 12th day of incubation, the dry weight reached 370 mg L-1 of O. quadripunctulata grown in standard medium corresponding to 340 mg L-1 in petrochemical effluent. The reduction amounted to 8.8% for cultures grown in wastewater compared to the standard medium.
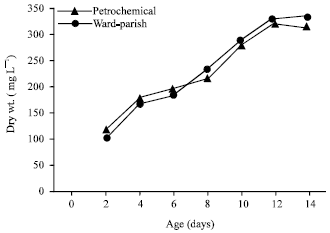 | |
| Fig. 3: | Growth of S. bijuga on petrochemical wastewater and Ward-Parish medium |
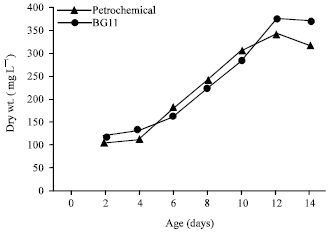 | |
| Fig. 4: | Growth of O. quadripunctulata on petrochemical wastewater and BG11 medium |
Pigments and protein content of S. bijuga and O. quadripunctulata: Table 1 shows the comparative analysis of chlorophyll a, carotenoid and protein content of S. bijuga grown in sterilized petrochemical effluent, sewage wastewater and standard medium after 12 days of incubation at 30±2°C. It was observed that the S. bijuga grown in sewage wastewater contained higher chlorophyll a, carotenoid and protein content with values reached 3.82, 1.90 and 182 μg mL-1 corresponding to standard medium (Table 1). Similarly S. bijuga grown in petrochemical effluent shows reduction in all content, 2.32, 1.33 and 119 μg mL-1 compared to standard medium, 2.79, 1.51 and 142 μg mL-1, respectively. Therefore, wastewater highly significantly increased the three growth criteria with 37.1, 24.2 and 28.2% (p<0.05) for chlorophyll a, carotenoids, protein content, respectively, over the control culture. The reduction of pigments showed cultures grown in petrochemical effluent than standard medium (16.8, 12.2 and 16.1%).
| Table 1: | Contents of chlorophyll a, carotenoid and protein (μg mL-1) of S. bijuga cultured in sterilized sewage and Petrochemical effluent wastewater after 12 days of incubation at 30±2°C |
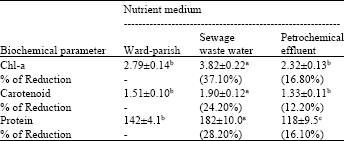 | |
| Data are presented as Mean±Standard deviation (n = 3). Different alphabets within the same row indicate significant differences at p<0.05 | |
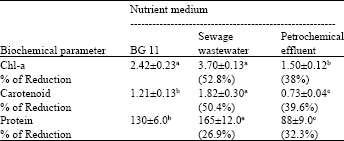
| Table 2: | Contents of chlorophyll a, carotenoid and protein (μg mL-1) of O. quadripunctulata cultured in sterilized sewage and Petrochemical effluent wastewater after 12 days of incubation at 30±2°C |
 | |
| Data are presented as Mean±Standard deviation (n = 3). Different alphabets within the same row indicate significant differences at p<0.05 | |
The results of chlorophyll a, carotenoid and protein content of O. quadripunctulata grown in sewage wastewater, petrochemical effluent and standard medium after 12 days of incubation at 30±2°C are presented in Table 2. The results showed that the wastewater was responsible for the increase of chlorophyll a, carotenoid and protein content of O. quadripunctulata to 3.7, 1.82, 165 μg mL-1 (p<0.05) corresponding to 2.42, 1.21 and 130 μg mL-1, respectively, in the BG11 medium. Sewage wastewater was significantly enhanced the three growth criteria of O. quadripunctulata by 52.8, 50.4 and 26.9% in chlorophyll a, carotenoid and protein, respectively.
The petrochemical effluent negatively affected pigments and protein content of O. quadripunctulata compared to BG11 medium. Thus, there was a highly significant in chlorophyll a, carotenoid and protein content reached 38, 39.6 and 32.3% below the control culture,, respectively.
Heavy metal removal from sewage wastewater and petrochemical effluents by S. bijuga and O. quadripunctulata: The capacity of S. bijuga and O. quadripunctulata cultivation to remove toxic heavy metals from sewage wastewater after 12 days of incubation has been presented in Table 3.The data reveal that the potentiality of removal differed according to the types of heavy metal and microorganism used.
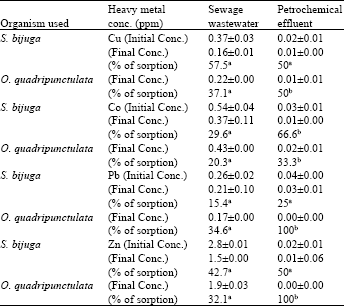
| Table 3: | The biosorptive capacity of S. bijuga and O. quadripunctulata culture to remove heavy metals from sewage wastewater and petrochemical effluent after 12 days of incubation |
 | |
| Data are presented as Mean±Standard deviation (n = 3). Different alphabets within the same row indicate significant differences at p<0.05 | |
It was obvious that the green algae used here exhibited high ability to remove heavy metals from sewage wastewater. S. bijuga was able to remove copper, cobalt and zinc from wastewater by 57.5, 29.6 and 42.7%, respectively but less content of lead was removed, 15.4%. While the removal efficiency was 37.1, 20.3 and 32.1% and high level of lead (p<0.05) was removed, 34.6% by O. quadripunctulata than S. bijuga.
The biosorptive capacity of the micro algae to remove heavy metals from petrochemical effluent was statistically highly significant (Table 3).The removal efficiency of copper, cobalt, lead and zinc reached 50, 33.3, 100 and 100%, respectively, in cultures of O. quadripunctulata compared with S. bijuga (50, 66.6, 25 and 50%).The relation between the decreases in heavy metal concentrations and the biosorbtive capacity of the cyanobacteria was highly significant (Table 3).
DISCUSSION
The growth of microalgae is indicative of water pollution since they respond typically to many ions and toxins. Blue-green algae are ideally suited to play a dual role of treating wastewater in the process of effective utilization of different constituents essential for growth leading to enhanced biomass production. Microalgae are efficient in the removal of nutrients from wastewater. Thus many microalgal species proliferate in wastewater due to the abundance of carbon, nitrogen and phosphorus that act as nutrients for the algae. Unicellular algae have shown great efficiency in the uptake of nutrients and have been found to show dominance in oxidation ponds (Rawat et al., 2011). Some of the wastewater being used here had excellent growth rates of S. bijuga (320 mg L-1) and O. quadripunctulata (441 mg L-1) grown in sewage wastewater even superior to those cultivated in synthetic medium. This may be due to the presence of available organic matter, phosphorus and nitrogenous compounds which support microbial growth. This observation is in agreement with (Sallal, 1986) who found that various types of filamentous and non-filamentous cyanobacteria were grown in sterilized treated and primary treated sewage effluents.
The growth of the S. bijuga in sterilized wastewater in some cases like those grown in petrochemical effluents and O. quadripunctulata grown in petrochemical effluents were relatively lower than those grown in standard medium (WP medium, 322 mg L-1 and BG11, 370 mg L-1) of the same inoculum size and under similar conditions and this may be due to the difference in the chemical composition between the wastewater and the well-balanced medium. This observation was similar to that of Lau et al. (1995) who mentioned that the growth of C. vulgaris grown in the primary settled sewage wastewater was lower than that grown in the commercial Bristol medium.
Furthermore, the rather high level of heavy metals which may limit the microbial growth, was considered another reason for the toxicity of wastewater (Kobbia et al., 1995). The biosorption mechanism can be considered as being dependent on the composition of the algal cell wall. Mostly, algal cell walls are made up of cellulose, polysaccharides, such as mannans, xylans and chitin (Rezaee et al., 2006; Okuo et al., 2006). These components, along with the proteins present, can provide acid-binding sites, such as amino, amine, hydroxyl, imidiazole, phosphate, carboxylates, thiols and thioesters and sulphate groups. Finally, the lower growth rates (315 and 340 mg L-1) may be due to the deficiency in one element or some elements of wastewater being used, like the deficiency in phosphorus content of petrochemical effluents. There are very few studies that look at the effects of refinery effluent or its components on algae. Saha and Konar (1985) used 90-day toxicity tests on phytoplankton. It was found that at the highest concentration tested (5.84% refinery effluent) the phytoplankton numbers decreased.
In some cases, the S. bijuga (3.82, 1.90 and 182 μg mL-1) and O.quadripunctulata (3.7, 1.82, 165 μg mL-1) of both grown in sterilized wastewater contained higher pigments and protein contents when compared with those grown in petrochemical effluent and standard medium. Microalgae (green and cyanobacteria) have been used to remove heavy metals from aqueous system, since they have a high capacity to accumulate dissolved metals and also improve the growth and pigment (Ting et al., 1989). The reduction of that content in petrochemical effluent treated micro algae may be due to the effect of effluent on the photosynthetic system by degrading the pigment content of the cells.
The metal sorption efficiency depends on the type of biosorbent, the availability and concentration of heavy metals. In this study, the percentage of reduction of Cu, Co, Pb and Zn was in the range between 37-50, 20.3-33.3, 34.6-100 and 32.1-100%, respectively from sewage wastewater and petrochemical effluent by ocillatoria culture. Prolonged contact with the metal-bearing solution, the living biomass is also capable of sequestering metal intracellularly. Oscillatoria have been shown to contribute significantly to the removal of heavy metals, such as Cu, Zn, Pb and Mn, that are discharged from a lead mine (Gale and Wixon, 1979). In O. anguistissima, copper and zinc binding was accompanied by release of large amounts of magnesium ions. The studies on Oscillatoria sp. showed its excellent copper biosorption qualities. The water bodies having high level of metal contamination supported growth of some major algal forms which accumulated substantial amount of metals in their tissues (Fereshteh et al., 2007). The metal absorption by S. bijuga were 60-50, 29.6-66, 15.4-25 and 42.9-50% from sewage and petrochemical effluents. Brady et al. (1994) found that Scenedesmus, Selenastrum and Chlorella sp. were capable of accumulating metals such as Cu and Pb with 67 and 98% efficiency. The growth inhibition in microalgae is related to the amount of heavy metal ions bound to the algal cell surface and also, to the amount of intracellular heavy metal ions (Nabizadeh et al., 2006).
In this study, it was observed that microalgae cultivated in sewage wastewater with high metal contents also accumulated higher metal contents. Das and Ramanujam (2011) reported that adsorption capacity, quantitative uptake and accumulation of Zn, Pb, Mn and Fe by Microspora quadrata. In case of O. quadripunctulata grown sterilized sewage wastewater, the present results showed that high concentrations of heavy metals did not affect the growth of the microalgae but it enhance its growth. This may be due to the resistance of the cyanobacterium to certain heavy metals. Decrease in growth with increasing concentrations of the petrochemical factory effluent was recorded by El-Dib et al. (1997). With reference to the heavy metal removal efficiency from wastewater and petrochemical effluent, by using both culture, better growth rate was shown at certain level, it may be due to the resistance or may be due to the low level of toxicity of effluents.
CONCLUSION
The present study, has demonstrated that microalgae biomass possesses the sorption capacity to remove heavy metal ions and other organic compounds. The investigation shows that S. bijuga and O. quadripunctulata are growing abundantly, hence they can absorb high amount of heavy metals from aqueous solutions. The growth of both species has supported wastewater as a growth medium compared to standard medium. The growth of microalgae in petrochemical effluent was reduced the growth by 2.2% (S. bijuga) and 8.8% (O. quadripunctulata) compared to the standard medium. The biosorption performance of S. bijuga was high in accumulating Cu (57.5%), Co (29.6%) and Zn (42.7%) than O. quadripunctulata. Whereas low amount of Pb accumulation was occured by S. bijuga. The microalgae wastewater treatment systems present a good option of biological tertiary treatment of sewage and petrochemical wastewater.
ACKNOWLEDGMENT
The study was supported by University Grant commission, UGC-SAP, New Delhi, funded for meritorious student’s project.
REFERENCES
- Azmat, R., F. Naz, T. Khanum and A. Hayat, 2005. Persistence of pathogens in heavy metals contaminated environment. Pak. J. Biol. Sci., 8: 235-238.
CrossRefDirect Link - Azmat, R., Uzma and F. Uddin, 2007. Biosorption of toxic metals from solid sewage sludge by marine green algae. Asian J. Plant Sci., 6: 42-45.
CrossRefDirect Link - Bradford, M.M., 1976. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem., 72: 248-254.
CrossRefPubMedDirect Link - Das, M. and P. Ramanujam, 2011. Metal content in water and green filamentous algae Microspora quadrata Hazen from coal mine impacted streams of Jaintia Hills District, Meghalaya, India. Int. J. Bot., 7: 170-176.
CrossRefDirect Link - El-Dib, M.A., H.F. Abou-Waly and A.M.H. Ei-Naby, 1997. Impact on fuel oil on the freshwater alga Selenastrum capricomutum. Bullet. Environ. Contam. Toxicol., 59: 438-444.
PubMed - Fereshteh, G., B. Yassaman, A.M.M. Reza, A. Zavar and M. Hossein, 2007. Phytoremediation of arsenic by macroalga: Implication in natural contaminated water, Northeast Iran. J. Applied Sci., 7: 1614-1619.
CrossRefDirect Link - Gale, N.L and B.G. Wixon, 1979. Control of Heavy Metals in the Lead Industry Effluents by Algae and Other Aquatic Vegetation. In: Management and Control of Heavy Metals in the Environment: International Conference, Aaseth, J., M. Astruc and R. Perry (Eds.). CEP Consultants Ltd., Edinburgh, UK., pp: 580-583.
- Han, X., Y.S. Wong, M.H. Wong and N.F.Y. Tam, 2007. Biosorption and bioreduction of Cr(VI) by a microalgal isolate, Chlorella miniata. J. Hazard. Mater., 146: 65-72.
CrossRefPubMedDirect Link - Haritonidis, S. and P. Malea, 1999. Bioaccumulation of metals by the green alga in Ulva rigida from Thermaikos Gulf, Greece. Environ. Pollut., 104: 365-372.
CrossRef - He, S. and G. Xue, 2010. Algal-based immobilization process to treat the effluent from a secondary wastewater treatment plant (WWTP). J. Hazard. Mater., 178: 895-899.
CrossRef - Huang, X.D., Y. El Alawi, J. Gurska, B.R. Glick and B.M. Greenberg, 2005. A multi-process phytoremediation system for decontamination of persistent Total Petroleum Hydrocarbons (TPHs) from soils. Microchemical. J., 81: 139-147.
Direct Link - Khorramabadi, G.S. and R.D.C. Soltani, 2008. Evaluation of the marine algae Gracilaria salicornia and Sargassum sp. for the biosorption of Cr (VI) from aqueous solutions. J. Applied Sci., 8: 2163-2167.
CrossRefDirect Link - Lau, P.S., N.F.Y. Tam and Y.S. Wong, 1995. Effect of algal density on nutrient removal from primary settled wastewater. Environ. Pollut., 59: 59-66.
CrossRefDirect Link - Mackinney, G., 1941. Absorption of light by chlorophyll solutions. J. Biol. Chem., 104: 315-322.
Direct Link - Nabizadeh, R., N. Kazem and S. Reza, 2006. Biosorption of lead (II) and cadmium (II) from aqueous solutions by protonated Sargassum sp. Biomass. Biotechnology, 5: 21-26.
CrossRefDirect Link - Okuo, J.M., S.B. Sanni and S.U. Aigbedion, 2006. Selective biosorption of heavy metal ions from aqueous solutions by pre-treated Nigerian fresh water algae. Trends Applied Sci. Res., 1: 83-90.
CrossRefDirect Link - Rawat, I., R.R. Kumar, T. Mutanda and F. Bux, 2011. Dual role of microalgae: Phycoremediation of domestic wastewater and biomass production for sustainable biofuels production. Applied Energy, 88: 3411-3424.
CrossRefDirect Link - Rezaee, A., B. Ramavandi, F. Ganati, M. Ansari and A. Solimanian, 2006. Biosorption of mercury by biomass of filamentous algae Spirogyra species. J. Boil. Sci., 6: 695-700.
CrossRefDirect Link - Stanier, R.Y., R. Kunisawa, M. Mandel and G. Cohen-Bazire, 1971. Purification and properties of unicellular blue-green algae (order Chroococcales). Bacteriol. Rev., 35: 171-205.
PubMedDirect Link - Wang, L., J. Peng, B. Wang and L. Yang, 2006. Design and operation of an eco-system for municipal wastewater treatment and utilization. Water Sci. Technol., 54: 429-436.
PubMedDirect Link








