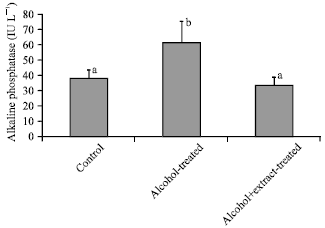
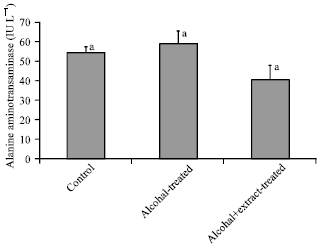
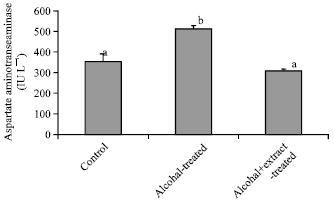
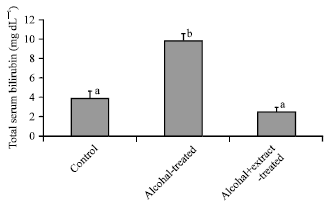
Research Article
Hepatotherapeutic Effect of Aloe Vera in Alcohol-induced Hepatic Damage
Department of Physiology, Ladoke Akintola University of Technology, Ogbomoso, Oyo State, Nigeria
R. E. Akhigbe
Department of Physiology, Ladoke Akintola University of Technology, Ogbomoso, Oyo State, Nigeria
O. S. Ishola
Department of Physiology, Ladoke Akintola University of Technology, Ogbomoso, Oyo State, Nigeria
E. A. Ashamu
Department of Anatomy, Ladoke Akintola University of Technology, Ogbomoso, Oyo State, Nigeria
O. T. Olayemi
Department of Anatomy, Ladoke Akintola University of Technology, Ogbomoso, Oyo State, Nigeria
G. E. Adeleke
Department of Biochemistry, College of Health Sciences, Ladoke Akintola University of Technology, Ogbomoso, Oyo State, Nigeria