Research Article
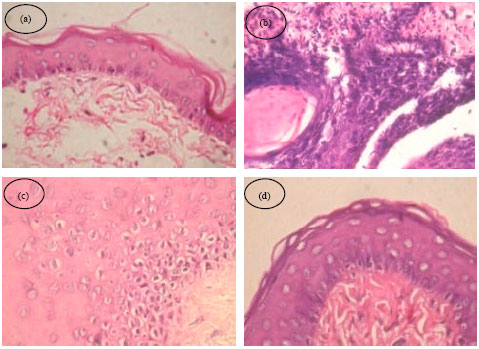
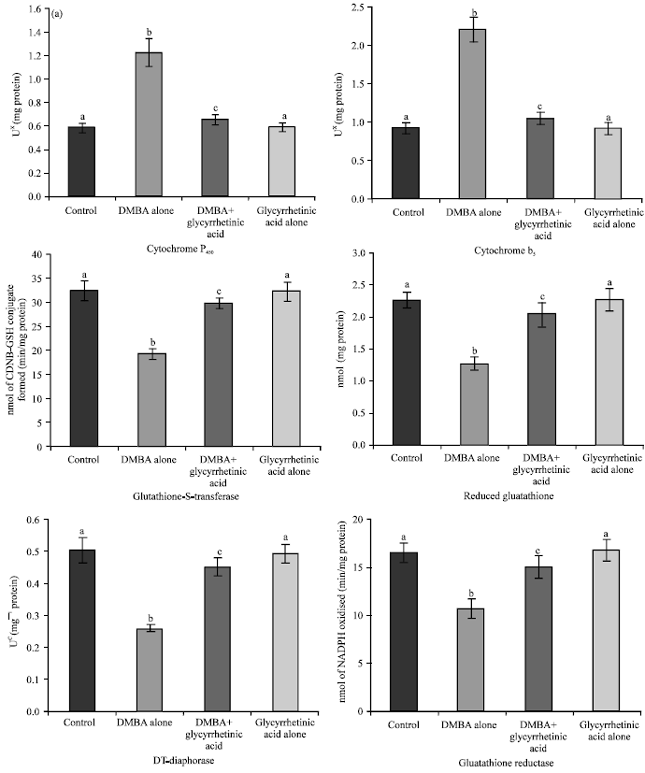
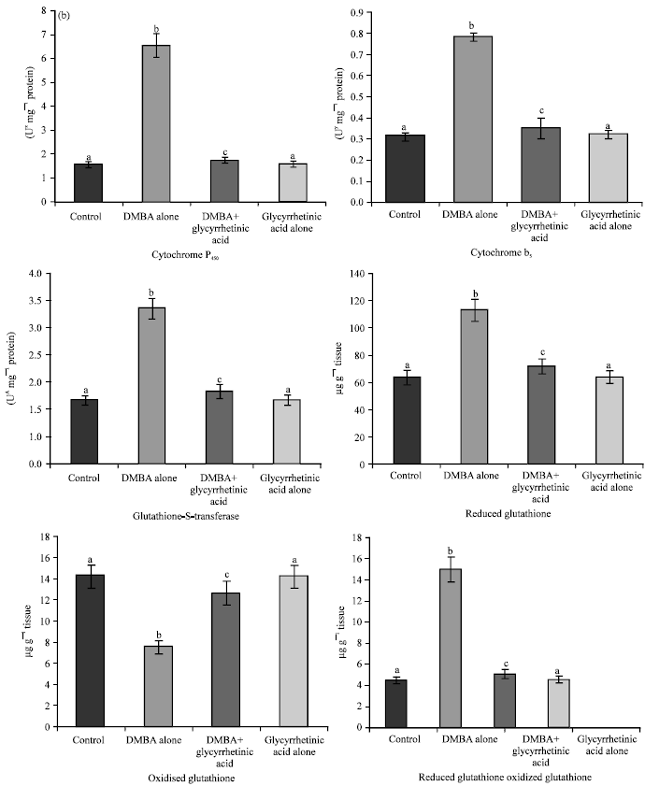
Chemopreventive Potential of 18β-glycyrrhetinic Acid: An Active Constituent of Liquorice, in 7,12-dimethylbenz(a)anthracene Induced Hamster Buccal Pouch Carcinogenesis
Department of Biochemistry and Biotechnology, Faculty of Science, Annamalai University, Annamalainagar-608 002, Tamil Nadu, India
P. Vishwanathan
Department of Pathology, Faculty of Medicine, Annamalai University, Annamalainagar-608 002, Tamil Nadu, India
S. Manoharan
Department of Biochemistry and Biotechnology, Faculty of Science, Annamalai University, Annamalainagar-608 002, Tamil Nadu, India