ABSTRACT
This study centers on development of the enzyme presentation as a cathode catalyst. Laccase enzyme is immobilized at nano meter scale in quaternary ammonium bromide salt treated Nafion polymers and assorted with high surface area carbon powders that are then deposited onto a solid support to create the cathode electrode. Optimization of the synthesis circumstances resulted in notably enhanced performance toward oxygen reduction reaction.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/pjbs.2010.611.612
URL: https://scialert.net/abstract/?doi=pjbs.2010.611.612
INTRODUCTION
An immobilized enzyme (with nanometer scale) is an enzyme which is attached to an inert, insoluble material (Legoy et al., 1979; Mansson et al., 1983). This can provide increased resistance to changes in conditions such as pH or temperature. It also allows enzymes to be held in place throughout the reaction, following which they are easily separated from the products and may be used again-a far more efficient process and so is widely used in industry for enzyme catalysed reactions. An alternative to enzyme nano immobilization is whole cell immobilization (Egwim and Oloyede, 2008; Tarek et al., 2007).
Biofuel cells are a good-looking choice fuel cell technology with the possible to enlarge moveable authority into uses that are not addressable by or are ill suited to conventional fuel cells or batteries (Addo et al., 2010). Akermin, in teamwork with the University of New Mexico, have established the feasibility of enhanced electronic relations between biocatalysts and engineered surfaces in order to harvest power and energy through biologic systems. These catalyst structures are used in biofuel cells (Gellett et al., 2010). Biofuel cells are different from straight fuel cells by the materials used to catalyze the redox reactions (Hideko and Marco, 1992). Rather than using valuable metals as catalysts, biofuel cells rely on biological catalysts recognized as enzymes, with an environmentally benevolent nature, to carry out the redox reactions. This study focuses on improvement of the enzyme performance as a cathode catalyst. For this target, Laccase enzyme is nano-immobilized in quaternary ammonium bromide salttreated Nafion polymers and mixed with high surface area carbon powders that are then deposited onto a solid support to create the cathode electrode (Moore, 2004). Beforehand demonstrated a laccase catalyzed air breathing biocathode as shown in Fig. 1.
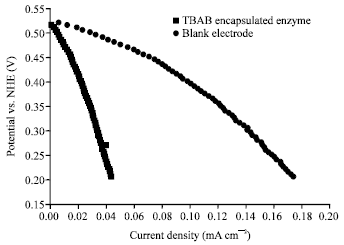 | |
| Fig. 1: | Air breathing biocathode half cell polarization curves of nano-immobilized enzyme 0.5 M potassium hydroxide |
MATERIALS AND METHODS
All chemical purchased from Merk Company. Experiment was carried out during 2008-2009 at amir-kabir laboratory. The Pt electrode was chosen as working electrode. In the complex inter-penetrating structure of a gas-diffusion air-breathing cathode, this frequently means providing specific transport trails at hierarchical scales for oxygen supply (through the gas phase), water withdrawal and proton access (through the liquid phase), while maintaining electrical (electronic) connectivity (through the solid phase). The enzyme catalyst is most efficiently placed at the tri-phase interface insuring efficient charge transfer and interaction with protons and oxygen leading to water formation. It has engaged in designing materials that will enable placement of the enzyme in such a hierarchical and interconnected matrix through a novel approach of templating of microemulsions (Fig. 2). TEM measurement was carried out by connection a TEM nano analyzer (ZAX model) to Pentium S.
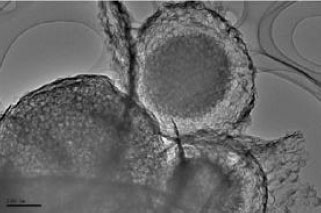 | |
| Fig. 2: | TEM microphotograph of the nano-structured non-Pt catalyst developed at UNM through templating of the precursors on microemulsion derived silica |
RESULTS AND DISCUSSION
Presentation of the cathode can be enhanced through straight electron transfer among the enzyme and electrode material (Akers et al., 2005).
The expansion of hierarchically-structured electro-catalysts and their supports can successfully address some of the present's confines of fuel cells. It is extremely attractive to amplify the loading of the enzyme on the electrode, which should lead to the increase of the obtainable power densities (and energy densities). One way to boost the current density is through designing the electrocatalyst material not only for high surface area but also with high amount of easy to get to three-phase sites. Porous structures can successfully minimize transport limitations, thus growing the convenience of the active sites by gas and electrolyte phases. Addressing the interactions of enzyme catalysts with the electrode interface can ease the electron transfer in sensible matters only if the materials design also answers the necessities for supporting the reaction outside the diffusion limited regime (Minteer et al., 2004). It was engaged in designing materials that will allow assignment of the enzyme in such a hierarchical and interconnected matrix through a new approach of templating of microemulsions as shown in Fig. 1. These new materials were made by templating on porous silica particles, formed by simultaneous templating with micelles and microemulsion droplets. Silica particles with bimodal pore distribution were then used as supports for pyrolizing non-Pt catalyst precursors. Morphology, composition and structure of templated cocatlyst and bedecked transition metal nano-phase were studied through extensive characterization by Scanning Electron Microscopy (SEM) and Transmission Electron Microscopy (TEM), X-Ray Diffraction (XRD) and X-ray Photoelectron Spectroscopy (XPS). Optimization of the synthesis conditions resulted in significantly improved performance toward oxygen reduction reaction, evaluated in rotating disk electrode and gas-diffusion electrode.
CONCLUSION
This study explains the integration of a new class of templated materials as co-catalysts for oxygen reduction as nano-immobilization supports for enzymes in biofuel cell cathodes. Silica particles with bimodal pore division were then second-hand as supports for paralyzing nano-Pt catalyst precursors. Optimization of conditions consequent in considerably enhanced presentation toward oxygen reduction reaction, assess in rotating disk electrode and gas-diffusion electrode.
REFERENCES
- Akers, N.L., C.M. Moore and S.D. Minteer, 2005. Development of alcohol/O2 biofuel cells using saltextracted tetrabutylammonium bromide/Nafion membranes to immobilize dehydrogenase enzymes. Elctrochimica Acta, 50: 2521-2525.
CrossRef - Hideko, Y. and M. Marco, 1992. NADH electrochemical sensor coupled with dehydrogenase enzymes. Anal. Lett., 25: 983-997.
CrossRef - Legoy, M.D., V.L. Garde, F. Ergan and D. Thomas, 1979. Study of immobilized α-steroid dehydrogenase in water-methanol media. Applied Biochem. Biotechnol., 4: 143-148.
CrossRef - Mansson, M.O., S. Nils and K. Mosbach, 1983. Site-to-site directed immobilization of enzymes with bis-NAD analogues. Proc. Natl. Acad. Sci. USA., 80: 1487-1491.
Direct Link - El-Banna, T.E., A.A. Abd-Aziz, M.I. Abou-Dobara and R.I. Ibrahim, 2007. Production and immobilization of α-amylase from Bacillus subtilis. Pak. J. Biol. Sci., 10: 2039-2047.
CrossRefPubMedDirect Link - Addo, P.K., R.L. Arechederra and S.D. Minteer, 2010. Evaluating enzyme cascades for methanol/air biofuel cells based on NAD+-dependent enzymes. Electroanalysis, 22: 807-812.
CrossRef - Gellett, W., M. Kesmez, J. Schumacher, N. Akers and S.D. Minteer, 2010. Biofuel cells for portable power. Electroanalysis, 22: 727-731.
CrossRefDirect Link








