Research Article
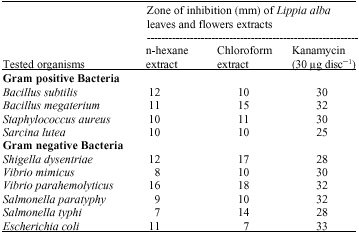
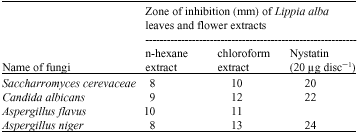
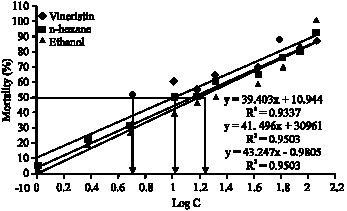
In vitro Antimicrobial and Cytotoxic Activities of Leaves and Flowers Extracts from Lippia alba
Department of Pharmacy, University of Southeast, Dhaka-1213, Bangladesh
M.H. Nur
Department of Pharmacy, University of Southeast, Dhaka-1213, Bangladesh
M.S. Amran
Department of Pharmaceutical Chemistry, Faculty of Pharmacy, University of Dhaka, Dhaka-1000, Bangladesh
M.I.I. Wahid
Department of Pharmacy, University of Rajshahi, Rajshahi-6025, Bangladesh
M. Ahmed
Department of Pharmacy, University of Rajshahi, Rajshahi-6025, Bangladesh