ABSTRACT
Using the scanning electron microscope, the gills of Schizothorax curvifrons have been studied after carrying out their primary fixation in 2.5% glutaraldehyde in 0.1 M sodiumcacodylate buffer (pH 7.3) for 24 h at 0-4°C and secondary fixation in 1-4% osmiumtetraoxide in 0.1 M sodiumcacodylate buffer (pH 7.3) for 1-2 h at 0-4°C. The gills are observed to comprise of gill arch, gill rakers having minute projections on their inner sides and gill filaments. The filaments originating from the gill arch are in a linear fashion and equidistant from each other, bearing secondary lamellae on both sides of the filament axis and each lamellae running parallel to the adjacent lamellae. The filament epithelium is found to be equipped with pavement cells, mucous cells and chloride cells. The pavement cells have smooth surface with sparse and irregular microridges defining their cell limits. The mucous cells are mainly found on the leading and trailing edges of the filament and the chloride cells are located close to the onset of secondary lamellae. The possible roles of these structures and cells in relation to respiration, water flow and feeding are discussed, setting thereby a platform for further studies in relation to pathology, pollution and stress conditions in aquaculture, natural and polluted environments.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/pjbs.2009.1511.1515
URL: https://scialert.net/abstract/?doi=pjbs.2009.1511.1515
INTRODUCTION
Regardless of lineage, the majority of fish species uses the gills as the primary site of aquatic respiration having complex organization that is similar in most teleosts and are multifunctional in nature. The gills form a highly characteristic feature of fishes and their presence has a marked effect on the anatomy and functioning of the rest of the animal. The supporting structure of gills in teleosts is the branchial arch, the next level of organization is the primary lamellae that finally supports the terminal secondary lamellae. The most superficial layer of the gill is made up of pavement cells (90%), chloride cells and mucous cells. The gross and fine structure of the gills of several teleosts have been extensively studied (Boyd et al., 1980; Kudo and Kimura, 1984; Moron and Fernandes, 1996; Carmona et al., 2004). The teleostean gills are involved in a number of functions such as: gas transfer, acid-base regulation and ion regulation. Further the gills are very sensitive to different environmental conditions and can undergo pathological changes which may be mediated by parasites including microbes and viruses as well.
The Snow trout, Schizothorax curvifrons locally called as Satter gad is a prized indigenous Schizothorax of Kashmir valley whose population is declining day by day. The fish appears to be the morphometrically and meristically most variable and valuable food species of the Kashmir vale but can always be recognized by the combination of large high scale count, high gill raker number and thin lips. The present investigation has been undertaken as effort to delimit what is known and unknown about this interesting organ, besides providing a base line for comparison with pathological and stress conditions in aquaculture and natural or polluted environment.
MATERIALS AND METHODS
Living specimens of normal and healthy Snow trout, Schizothorax curvifrons (25-30 cm in length, body weight 300-350 g) were collected from the world famous Dal Lake of the paradise vale. The fishes were killed on the spot with a blow to the head. The head was immediately decapitated to remove the gills. The excised gills were rinsed in physiological saline to remove excess mucin and other adhered particles. After rinsing the gill samples were fixed in 2.5% glutaraldehyde in 0.1 M sodiumcacodylate buffer (pH 7.3) for 24 h at 0-4°C (Primary fixation). Following primary fixation the samples were transferred to 0.1 M sodiumcacodylate buffer for 24 h and post fixed in 1-4% osmiumtetraoxide in 0.1 M sodiumcacodylate buffer (pH 7.3) for 1-2 h at 0-4°C (Secondary fixation). The post fixed samples were then dehydrated in increasing concentrations of ethanol (30-100%) and critical point dried with carbon dioxide. The gill samples were then mounted on the metal stubs with a paste of colloidal sliver paste and coated with a thin conductive film of gold in a sputtering coater and examined under LEO 435 VP scanning electron microscope at All India Institute of Medical Sciences (AIIMS) New Delhi India.
RESULTS
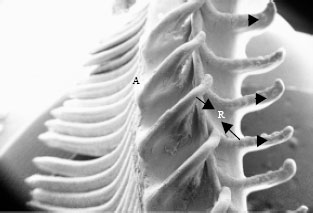
In Schizothorax curvifrons the gross structure of the gills are similar to those found in other teleosts. Each gill is found to consist of gill arch, gill rakers and gill filaments (Fig. 1). Arising from the gill arch are a number of gill filaments which are arranged in a single row and are equidistant from each other giving it a leaf like structure (Fig. 2). The shape of the filaments is of very filamental type but the length exceeds the breadth. The secondary lamellae are also observed to be projecting at right angles to the long axis of the filament each lamellae lying parallel to the adjacent lamellae (Fig. 3). A numbers of rakers (21-28) are observed to radiate from both sides of the gill arch on the opposite sides of the gill filament, the inner sides of the rakers are equipped with minute projections (Fig. 4). The outer most layer of the filament consists mainly of pavement cells with mucous cells and chloride cells spread between them (Fig. 5). The chloride cells are found close to the onset of secondary lamellae and most of the mucous cells are found on the leading and trailing edges of the filament.
 | |
| Fig. 1: | Scanning electron micrograph of the gill showing gill rakers (R), gill arch (A) and filaments (F). X200 |
 | |
| Fig. 2: | Gill filaments (F) with leaf like lamellae on the upper and lower sides. X200 |
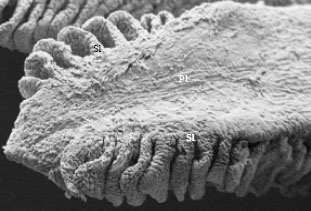 | |
| Fig. 3: | SEM of an individual filament showing the parallel arrangement of the secondary lamellae (Sl) on the primary lamellae (Pl). X500 |
 | |
| Fig. 4: | Scanning electron micrograph of gill showing distinct rakers (R) with minute projections ( |
 | |
| Fig. 5: | SEM of filament epithelium showing pavement cells (PVC), chloride cells (CC), mucous cells (MC) and microridges (→). X959 |
The pavement cells are polygonal in shape having almost smooth surface with sparse and irregular microridges and cell limits are well defined by large and circular microridges.
DISCUSSION
Scanning electron microscopic observations on the surface ultrastructure of the gills of Schizothorax curvifrons reveal the presence of well developed and compactly organized primary filaments which radiate from the gill arches. Both sides of the primary lamellae are noted to be lined up by secondary lamellae as also reported earlier in other teleosts (Kudo and Kimura, 1984; Ojha et al., 1987; Arellano et al., 2004). The gill filaments and their secondary lamellae represent two general types of epithelium (Laurent and Dunel, 1980; Laurent, 1984).
Filaments or the primary lamellae constitute the most prominent respiratory structures of the gills. Laurent (1984) stated that the shape of the gill filaments in general varies from being very filamental to fairly stubby structures. However, only filamental type of gill filaments are observed in the present study though the length exceeds the breadth. The filamental type of filaments may probably help in circulating more and more water through them and may also reduce the diffusion distance between the blood and the respiratory water. According to Hughes (1966), the number of filaments do not increase so markedly in adult fishes as during the juvenile growth period, although each filament exhibits significant increase in the length as the fish grows. This leads to an increase in the total length of all the filaments, which is an important morphometric dimension used in calculating the gill area. In Schizothorax curvifrons the gill filaments observed are long and narrow projections lateral to the gill arch that taper at their distal ends. The secondary lamellae are found evenly distributed along the length of the filament and are perpendicular to the long axis of the filament besides being parallel to each other. This is in conformity with the findings of Kudo and Kimura (1984) and Evans et al. (2005). The lamellae constitute the most important units of the gill system from the point of view of gas exchange (Laurent, 1984). According to Evans et al. (2005), the lamellae not only dramatically increase the surface area of the gill filament epithelium and result in small diffusion distance between the blood that perfuses each lamellae and the respiratory water but are also well suited for diffusive loses or gains of ions and water to or from the environment. From a design point of view, lamellae are required to have a large surface area where gas exchange can be facilitated without any excessive exchange of ions and water. A close contact between water and blood must be achieved so that oxygen uptake can occur in a limited period during which the water and blood are passing the lamellae.
Ojha et al. (1987) observed two types of gill rakers in the gills of freshwater mullet, Rhinomugil corsula and single type of gill rakers in Sicamugil cascasia. However, during the present study, only single type of gill rakers are noticeable which may reflect the variety of food passing through them and the selection of a particular type of food. The gill rakers are long and slender bearing minute projections on the inner side. These projections were also observed by Ojha et al. (1987) on the first type of gill rakers of the gills of Rhinomugil corsula. The well developed gill rakers and their specific orientation may possibly be related to the herbivorous nature of the fish studied. The densely packed rakers aid in the detection and capturing of food flowing through water. The presence of microridges on the gill rakers can be associated with the holding of mucous secreted by the goblet cells. The mucous serve to increase the efficiency of gill rakers by cleaning the surface and exposing the chemoreceptors for the detection of food and chemical characteristics of ambient water. Moreover, the arrangement and density of gill rakers are specific for different fishes and may be a guiding factor for determining food and feeding habits and the taxonomic status of the fish.
The filament epithelium of teleosts consists of pavement cells, mucous cells and chloride cells (Morgan and Tovell, 1973; Kendall and Dale, 1979; Fernandes and Martins, 2001) as is also evident from the present investigation. The major surface area of the gill filament is covered by pavement cells, which are largely considered to play a passive role in gill physiology and are significant or gaseous exchange. The microridges are noted to be present in between the pavement cells defining their cell boundaries. The presence of microridges on the apical membrane of pavement cells seem to increase the functional surface area of the epithelium and may also play a role in anchoring mucous to the surface (Hughes and Wright, 1970; Lewis, 1979; Fernandes and Martins, 2001; Evans et al., 2005). According to Olson and Fromm (1973), the nature of the microridges varies in fish species but no correlation between microridge structure and the physiology and ecology has yet been established.
The mucous cells are more or less similar to those found in the gills of other teleosts (Laurent, 1984; Fernandes and Martins, 2001). The mucous secreted by the mucous cells have different roles such as control of infection, prevention of dehydration, ion regulation and ion diffusion (Handy et al., 1989; Diaz et al., 2001). In addition, the mucous of the gill epithelia is also involved in swimming and defense against pollutants and parasites (Fletcher, 1978). Schizithorax curvifrons is a freshwater fish living generally in ion poor waters. The released mucin plays a significant role in the prevention of loss of ions.
The chloride cells in the gills of the fish studied are found close to the onset of secondary lamellae. Similar observations have been made by Fernandes and Martins (2001) with respect to the gills of Hypostomus plecostomus. Franklin and Davison (1989) while working on the scanning electron microscopy of the gills of freshwater adopted sockeye salmon, Oncorhynchus nerka, have reported two morphologically different chloride cells on the afferent surface of the gill filament. However, during the present study, only single type of chloride cells are noted to be present on the gill epithelium. The occurrence of single type of chloride cells is possibly due to the fact that the Schizothorax curvifrons is generally living in cold and ion poor waters Teleostean chloride cells seem to play an important role in ionic and osmotic regulation (Karnaky, 1986). According to Evans et al. (2005) chloride cells occupy a much smaller fraction of the branchial epithelia, but they are considered to be the primary active sites of physiological processes in the gills.
ACKNOWLEDGMENTS
The authors not only appreciate the excellent assistance of Dr. T.C. Nag (Associate Prof. Department. of Anatomy AIIMS) but also his helpful suggestions and sharing of knowledge on the gills throughout this study.
REFERENCES
- Boyd, R.B., A.L. DeVries, J.T. Eastman and G.G. Pietra, 1980. The secondary lamellae of the gills of cold water (high latitude) teleosts: A comparative light and electron microscopic study. Cell. Tissue Res., 213: 361-367.
Direct Link - Carmona, R., M. Garcia-Gallego, A. Sanz, A. Domezain and M.V. Ostos-Garrido, 2004. Chloride cells and pavement cells in gill epithelia of Acipenser naccarii: Ultrastructural modifications in seawater-acclimated specimens. J. Fish. Biol., 64: 553-566.
CrossRefDirect Link - Diaz, A.O., A.M. Garcia, C.V. Devincenti and A.L. Goldemberg, 2001. Mucous cells in-Micropogonias furnieri gills: Histochemistry and ultrastructure. Anat. Histol. Embryol., 30: 135-139.
Direct Link - Evans, D.H., P.M. Piermarini and K.P. Choe, 2005. The multifunctional fish gill: Dominant site of gas exchange, osmoregulation, acid-base regulation and excretion of nitrogenous waste. Physiol. Rev., 85: 97-177.
CrossRefDirect Link - Fernandes, M.N. and S.A.P. Martins, 2001. Epithelial gill cells in the armored catfish, Hypostomus plecostomus (Loricariidae). Rev. Braz. Biol., 61: 69-78.
PubMed - Franklin, C.E. and W. Davison, 1989. SEM observations of morphologically different chloride cells in freshwater adopted sockeye salmon, Oncorhynchus nerka. J. Fish Biol., 34: 803-804.
CrossRef - Hughes, G.M., 1966. The dimensions of fish gills in relation to their function. J. Expt.-Biol., 45: 177-195.
Direct Link - Hughes, G.M. and D.E. Wright, 1970. A comparative study of the ultrastructure of the water-blood pathway in the secondary lamellae of teleost and elasmobranch fishes-benthic forms. Z. Zelloforsch. Mikrosk. Anat., 104: 478-493.
PubMed - Karnaky, K.J., 1986. Structure and function of the chloride cells of Fundulus heteroclitusand other teleosts. Am. Zool., 26: 209-244.
CrossRef - Laurent, P. and S. Dunel, 1980. Morphology of gill epithelia in fish. Am. J. Physiol., 238: 147-159.
PubMed - Lewis, S.V., 1979. A scanning electron microscopic study of the gills of the air breathing catfish Clarias batrachus L. J. Fish Biol., 15: 381-384.
CrossRef - Morgan, M. and P.W. Tovell, 1973. The structure of the gill of the trout Salmo gairdneri (Rich.). Z. Zellforsch Mikrosk Anat., 142: 147-162.
PubMed - Moron, S.E. and M.N. Fernandes, 1996. Pavement cell ultrstructural differences on Hoplias malabaricus gill epithelia. J. Fish Biol., 49: 357-362.
Direct Link - Ojha, J., A.K. Mishra and J.S.D. Munshi, 1987. Interspecific variations in the surface ultrastructure of the gills of freshwater mullets. Ichthyol. Res., 33: 388-393.
CrossRef - Olson, K.R. and P.O. Fromm, 1973. A scanning electron microscopic study of secondary lamellae and chloride cells of rainbow trout (Salmo gairdneri). Z. Zellforsch. Mikrosk. Anat., 143: 439-449.
PubMed









Sumaira Reply
Dear sir,
As a research scholar i am highly impressed by this work and in real sense it has laid down the platform for future studies in cold water fisheries.
Dr. I.H.Mir
Thanks