Research Article
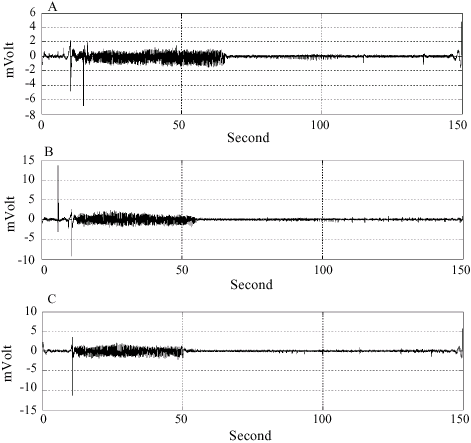
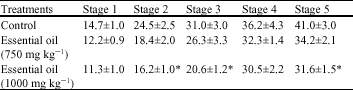
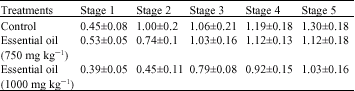
The Effect of Rosa damascena Essential Oil on the Amygdala Electrical Kindling Seizures in Rat
Department of Biology, Faculty of Sciences, Ferdowsi University of Mashhad, Iran
Ali Moghimi
Department of Biology, Faculty of Sciences, Ferdowsi University of Mashhad, Iran
Hassan Rakhshandeh
Department of Physiology and Pharmacology, Qaem Hospital, Mashhad University of Medical Sciences, Iran
Hamid Ejtehadi
Department of Biology, Faculty of Sciences, Ferdowsi University of Mashhad, Iran
Masoumeh Kheirabadi
Department of Biology, Faculty of Sciences, Ferdowsi University of Mashhad, Iran