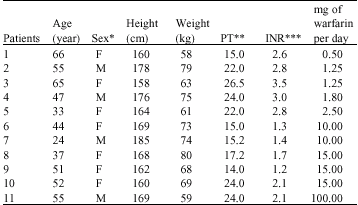
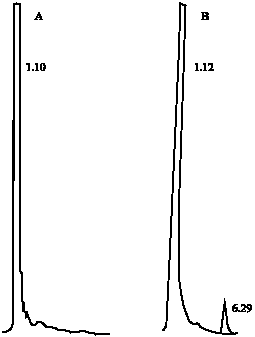
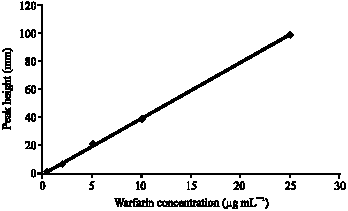
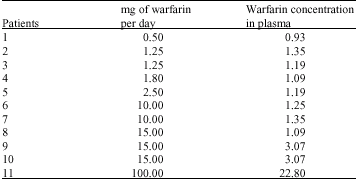
Research Article
Assaying of Warfarin in Iranian Warfarin Resistance Patients Blood by HPLC
Faculty of Pharmacy, Tehran University of Medical Sciences, Tehran, Iran
P. Ghadam
Department of Biochemistry, Faculty of Science, Alzahra University, Tehran, Iran
R. Sharifian
Department of Hematology, Imam Hospital, Tehran, Iran
F. Sadeghian
Department of Biochemistry, Faculty of Science, Alzahra University, Tehran, Iran