Research Article
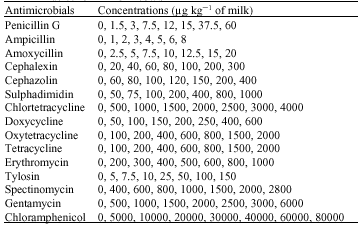
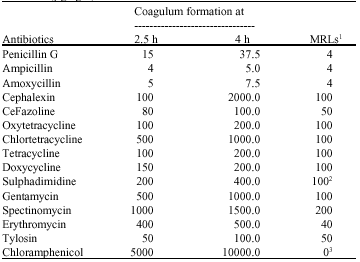
The Detection Limits of Antimicrobial Agents in Cow`s Milk by a Simple Yoghurt Culture Test
Department of Food Hygiene and Technology, Ferdowsi University of Mashhad, P.O. Box 91775-1793, Mashhad, Iran
A. Bahrainipour
Department of Food Hygiene and Technology, Ferdowsi University of Mashhad, P.O. Box 91775-1793, Mashhad, Iran