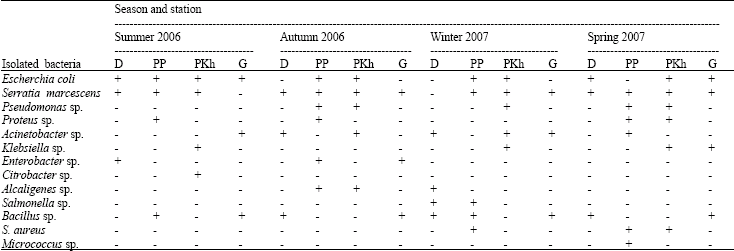
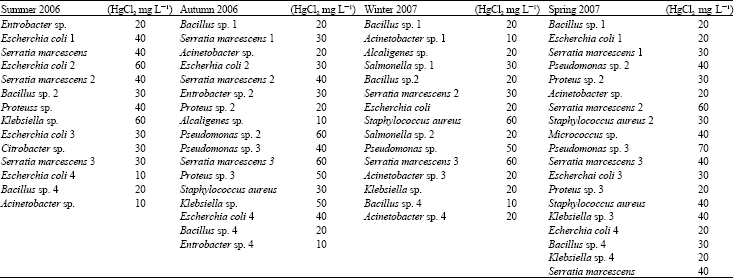
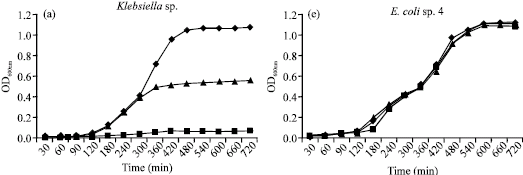
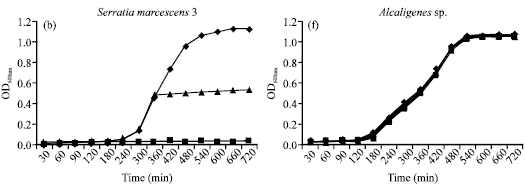
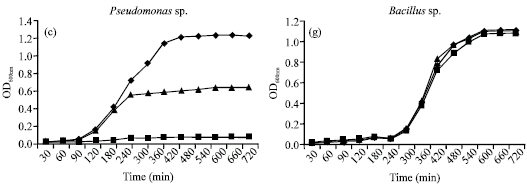
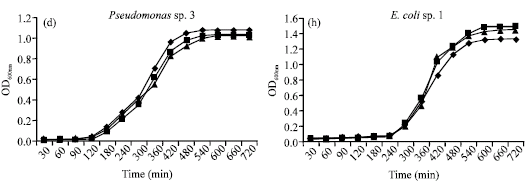
Research Article
Growth Pattern of Hg Resistant Bacteria Isolated from Kor River in the Presence of Mercuric Chloride
Department of Microbiology, Islamic Azad University of Jahrom, Fars Province, Jahrom, Iran
N. Mirzaei
Department of Microbiology, Islamic Azad University of Jahrom, Fars Province, Jahrom, Iran