ABSTRACT
The study of vitelline glands of digenean Haploporus lateralis, indicates that the glands consist of vitelocytes in various stages of maturation. The cytoplasmic process of nurse cells which surrounded the vitelline lobes, intrude between vitelocytes and surround them. Immature vitelline cells are undifferentiated, they possess small amount of cytoplasm and large nucleus. The synthesis of shell globules begin in the maturing vitelline cells. Increasing numbers of vitellocytes reach develop of mature vitelline cells. The cytoplasm of mature vitelline cells is filled with shell globules. Mature vitelline cells usually found in the center of vitelline lobe. They release into the vitelline duct and vitelline reservoir. The vitelline reservoir and duct are lined with a syncytial epithelium. Basement membrane is present between nurse cells and vitelline cells.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/pjbs.2008.113.117
URL: https://scialert.net/abstract/?doi=pjbs.2008.113.117
INTRODUCTION
There has been agreed for many years that in tromatodes the Mehlis` gland provides the egg shell, whereas, the vitelline cells provide the nutrient for developing embryo. An additional role for the vitelline gland which of producing the shell materials has been reported. Some studies have been performed on the fine structure on the vitellaria on the digenean including the work of Tulloch and Shapiro (1957), Bjorkman and Thorsell (1963), Irwin and Maguire (1979), Holy and Wittrock (1986) and Erasmus (1973) and they have reported that in some primitive species of digenean (with extensive vitelline gland) many intact vitelline cells enclosed with the ovum in the large egg, whereas, in some advanced species digenean like Microphayllidae two vitelline cells enclosed the shell formation. Some primitive digenetic families including Hemiruidae and Haploporidae, possesses small compact vitelline gland (Smyth and Clegg, 1959). Although Haploporus lateralis, possesses small vitelline gland, produces large number of eggs but small in size. Grant et al. (1977) and Irwin and Threadgold (1972) reported that the vitelline gland produce the vitelline globules, which some of them produce the shell and some provide nutrient for the developing embryo within the egg. Bjorkman and Thorsell (1963) showed that shell globules are formed within the vitelline cells. The ultra structure, vitellgenesis and associated ducts including, digenean Maritrema linguilla have been studied by Hendow and James (1989) and monogenean, Metamicrocotyla macrocauta, by Baptista-Farias (1998). Levran et al. (2007) reported the ultrastructural and histochemical in tapeworm, Paraechinophallus japonicus.
The role of vitelline gland in the egg-shell formation in digenean trematodes including Dicrocoelium dedriticum, have been investigated by Grant et al. (1977), Schistosoma mansoni, by Wells and Cordingley (1991), Haploporus benedenii by Sampour (2006) and in cestodes, Diplocotyle olrikii, by Poddubnaya et al. (2005).
In H. lateralis, the vitellogenesis occurs in the vitelline gland, mature vitelline cells leave the vitelline lobe and reach the central chamber via the vitelline duct and are enclosed in egg shell production.
In the present study, cytological changes in the vitellaria in Haploporus lateralis, have been studied with light and transmission electron microscopy, during the development of vitelline cells.
MATERIALS AND METHODS
Adult worms were obtained from the intestine of marine fish Mugil auratus. The investigation carried out in the university of Luristan, Khoramabadm, Iran, during years 2004-2005. The specimens were fixed in 70% ethanol, identification was facilitated by staining Gower`s Carmine or Fast Red B Salt. Several specimens of H. latralis, were studied with light microscope and several specimens were studied with transmission electron microscope. Thick sections were sliced at 0.5 μm and stained with 1% toluidine blue and observed under light microscope. The specimens were fixed in 5% gutaraldehyde, in sodium cacodylate at pH 7.2 buffer for Transmission Electron Microscopy (TEM) and post fixed in 1% osmium tetroxide (OsO4). The specimens were passed through graded acetone solution and embedded in Epon resin (Epon 812) which degassed in a vacuum chamber and polymerized at 60°C. Semithin and ultrathin sections (80 nm) were cut with a Huxley ultramicrotome. Ultrathin sections were mounted on uncoated copper grids, double stained with uranyl acetate in fresh methanol, destained through methanol series, oven dried at an oven at 60°C, post stained in lead nitrate and trisodium citrate. The grids were rinsed in distilled water and viewed in a Joel 1200 EX transmission electron microscopy at 60-80 KV.
RESULTS
The vitelline gland of Haploporus lateralis is represented by two small symmetrical compact lobes. The vitellaria is located in the mid-body behind the acetabulum close to the testis. Each vitelline lobe connected the vitelline reservoir by a short vitelline duct. The vitellin reservoir tends to the central chamber by a common duct. The central chamber is surrounded by cells of Mehlis gland. The vitelline gland produces vitellocytes, which are observed in each lobe at different stages of maturation (Fig. 1). The cells are distributed any where within lobes. The vitellaria is surrounded by nurse cells, which contain glycogen, the nurse cell`s cytoplasmic processes penetrated deeply into the vitelline gland ramify between and surround the developing vitellocytes (Fig. 2). There is a prominent basement membrane between nurse cells and vitelline cells (Fig. 2). Immature vitelline cells are irregular in shape with a prominent central nucleus, which occupied most volume of the cell. The nucleus often possesses a single nucleolus and some conspicuous heterochromatin dense, which are scattered in the nucleoplasm (Fig. 2). Small number of mitochondria which have distinct cristae and some granular endoplasmic reticulum and abundant free ribosomes are present in the cytoplasm of immature cells. Some of the ribosome`s attached the outer nuclear membrane. Immature vitelline cells lack the characteristic shell globules. In some cases Golgi complexes are difficult to find. During immature vitelline cells differentiation into developing vitelline cells, they involved some changes. As the cells begin maturation, they increased in size and number. Both the cytoplasm and nuclear increased in volume at this stage of development, but the nuclear/cytoplasmic ratio decreased. The nucleus contains some clumps of heterochromatin (Fig. 2), in which some of them close to the nuclear membrane. The nucleoplasm becomes more denser than that of immature cells. Maturing vitelline cells contain small Golgi complexes with saccullr and vesicles in different size and granular endoplasmic reticulum (Fig. 5). The granular endoplasmic reticulum is observed in the cytoplasm, usually adjacent to the outer nuclear membrane. There are few lipid droplets and some
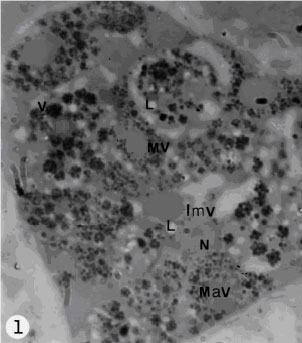 | |
| Fig. 1: | Arrangement of vitelline cells in the vitelline lobe at different stages of maturation. The gland is surrounded by nurse cells. Note immature vitelline cells(Imv); maturing vitelline cell (Mv); mature vitelline cell (Mav); L, lipid droplet; N, nucleus; v, vitelline globules |
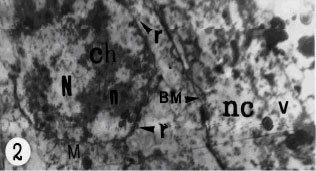 | |
| Fig. 2: | Transmission electron micrograph of immature vitelline cells with large nucleus (N) and little rate of cytoplasm; n, nucleoli; ch, heterochromatin in the nucleus; r, free ribosome in the cytoplasm and some of ribosome (r, arrow head) associated with the outer nuclear membrane. Basement membrane (BM) is present between nurse cells (nc) and vitelline gland; v, vacuole of nurse cells |
mitochondria with cristae in the cytoplasm of cells (Fig. 3). Free ribosomes are scattered in the cytoplasm. The vacuoles containing glycogen are present in the cytoplasm of maturing vitelline cells (Fig. 6). The maturing vitelline cells involved production of shell globules.
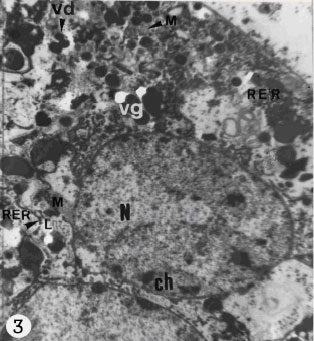 | |
| Fig. 3: | Transmission electron micrograph of maturing vitelline cells. Note N, nucleus; M, mitochondria; ch, hetrochromatin in the nucleus; RER, rough endoplasmic reticulum; vg, vitelline granules, which appears for the first time at this stage of maturation; L, lipid droplets; vd; vitelline droplets |
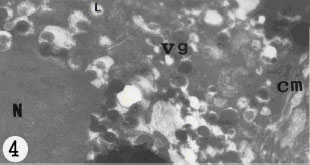 | |
| Fig. 4: | Electron micrograph of mature vitelline cell. The cell is limited by cell membrane (cm). The cytoplasm occupied with vitelline granules (vg); L, lipid droplets are in the cytoplasm of cell. Note N, nucleus |
Rough endoplasmic reticulum; associated with micro vesicle containing electron-dense materials or small droplets. They are also close to the Golgi complexes (Fig. 3, 5) These appear to form singly, then aggregate, increasing in electron density and increasing in size to produce larger globules (Fig. 5, arrows head, 1, 2, 3) which obtained their maximum size. The vitelline globules are also increased in number. However, the vitelline globules
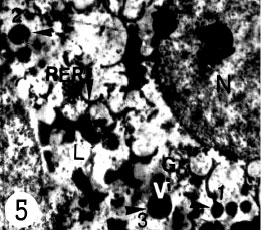 | |
| Fig. 5: | Electron micrograph of vitelline cell indicating development of vitelline granules (v), they appears as small electron dense materials first (arrow head, 1) then they increasing electron density and increasing in size (arrow head, 2) to produce larger globules (arrow head, 3). Then they arranged in clusters |
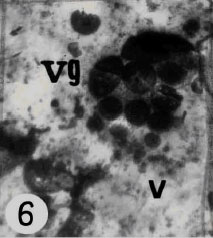 | |
| Fig. 6: | Electron micrograph of vitelline granules (vg) of vitelline gland arranged in clusters usually occupied the cytoplasm of cell. Note Vacuoles (v) contains glycogen are observed in the cytoplasm of cells |
are bounded, they are arranged in clusters in various sizes (Fig. 6), in some clusters the globules are angular, in outline, this causes them associated with each other within the clusters. In some cases the cytoplasm is occupied by clusters of shell globules (Fig. 3), the other parts of cytoplasm are filled with cell organelles. The mature vitelline cell possesses plasma membrane (Fig. 4). The cytoplasm of mature vitelline cells usually filled with the large rounded shell globules (Fig. 4). The nucleu contains nucleolus and patches of heterochromatin. Small lipid droplets and few mitochondria are present in the cytoplasm. The vitelline cells are released from the vitelline lobes and pass down the vitelline duct and vitelline reservoir. The vitelline cells discharged their contents as shell globules into the lumen of central chamber, where they participate to shell formation. Two short collecting vitelline ducts connected the vitelline lobes to the vitelline reservoir. A short vitelline duct runs posteriorly from the vitelline reservoir join the proximal of central chamber. The vitelline duct and vitelline reservoir are lined with a syncytial epithelium which is underlined by a circular muscle layer.
DISCUSSION
Information obtained from this study, would suggest that in Haploporus lateralis, the vitellaria consists of two compact lobes which produced the vitelline cells.
The general structure of vitelline cells of H. lateralis corresponds to that found in other digeneans.
The ultra structural evidence obtained from the vitelline cells development in H. lateralis is similar to that found in primitive species of digenean. In primitive species and H. lateralis, many healthy vithelline cells leave the vitelline gland and migrate to the central chamber, where they take part the egg shell formation, while in advanced species of digenean two vitelline cells are enclosed the egg shell formation in the central chamber.
In H. lateralis, as in other digenea (Martinez-Alos et al., 1993; Holy and Wittrock, 1986), three stages of developing vitelline cells, including immature, maturing and mature cells, are found. Immature cells are early cells of differentiating. Differentiation of immature vitelline cells into maturing cells, involved in increasing the volume and density of the cytoplasm and development of abundant granular endoplasmic reticulum, Golgi complex and production of dense droplets of shell globules (Halton et al., 1974). Maturing cells show the beginning of shell globule production, mature vitelline cells are characterized with vitalline granules which occupied throughout the cytoplasm. The granules in the mature cells are presented with larger size. The vitelline globules in the maturing or mature vitellocytes of H. lateralis are apparently equivalent to those displayed in the vitellocytes of other trematodes. Thulin (1982) reported that the mentioned droplets represent the source of vitelocytes. Shell protein is apparently produced by rough endoplasmic reticulum and packed with Golgi complex (Irwin and Threadgold, 1970; Hanna, 1976; Holy and Wittrock, 1986), they condense in small body, coalesce and increasing in size (Hendow and James, 1989).
In H. lateralis, like more primitive digenean species, healthy mature vitelline cells, possess cytoplasm with healthy organelles and healthy nucleus contains nucleolus, vitelline globules arranged in clusters, occupied most the cytoplasm and the cell membrane surrounded the cells. In this position, the cells break away from the vitelline lobes and pass into the vitelline duct. In contrast, in advanced digenean species, such as Mariterma linguilla (Hendow and James, 1989), in mature vitelline cells, the nucleus and cytoplasm degenerate and the cells contains only shell globules which plasma membrane surrounded them (Hendow and James, 1989), neither cytoplasm nor nucleus is present. In H. laterais the function of vitelline cells, as in other trematodes, included in production of shell globules and represent nutritive materials for the developing larva. The presence of mitochondria contain cristae in the vitelline cells, probably provide energy for cells activities.
Halton et al. (1997) reported that in trematodes, the nerve plexuses associated with reproductive system innervated different ducts of female organ including uterus, vitelline duct, oviduct and ootype. The contraction of neuro-muscular regulates actions of female reproductive duct (Poddubnaya et al., 2005). The passing of vitelline cells must be facilitated by peristalsis (Hendow and James, 1989).
The above mentioned concerning the structure and development of the vitelline cells of H. lateralis, is supported by micrographs obtained from this investigation.
REFERENCES
- Baptista-Farias, M.F.D., 1998. Ultrastructural observations of the vitelline cells of Metamicrocotyla macracautha (Monogenea: Microcotylidae). Memorias Instituto Oswaldo Cruz, 93: 543-548.
Direct Link - Bjorkman, N. and W. Thorsell, 1963. On the fine morphology of the formation of egg- shell globules in the vitelline gland of the liver fluke (Fasciola hepatica, L.). Exp. Cell Res., 32: 153-156.
PubMedDirect Link - Erasmus, D.A., 1973. A comparative study of the reproductive system of mature, immature and unisexual female Schistosoma mansoni. Parasitology, 67: 165-183.
PubMedDirect Link - Halton, D.W., S.D. Stranock and A. Hardcastle, 1974. Vitelline cell development in Monogenean parasite. Z. Parasitenk, 45: 45-61.
CrossRefDirect Link - Hanna, R.E.B., 1976. Fasciola hepatica: A light and electron autoradiographic study of shell protein and glycogen synthesis by vitelline follicles in tissue slices. Exp. Parasitol., 39: 18-28.
PubMedDirect Link - Hendow, H.T. and B.L. James, 1989. Ultra structure of vitellarium, vitellogenesis and associated ducts in Maritrema linguilla (Digenea: Microphallidae). Int. J. Parasitol., 19: 489-497.
Direct Link - Holy, J.M. and D.D. Wittrock, 1986. Ultrastructure of the female reproductive organs (ovary, vitellaria and Mehlis` gland) of Halipegus eccentricus (Trematod: Derogenidae). Can. J. Zool., 64: 2203-2212.
Direct Link - Irwin, S.W.B. and L.T. Threadgold, 1970. Electron microscope studies on Fasciola hepatica. VIII. The development of the vitelline cells. Exp. Parasitol., 28: 399-411.
PubMedDirect Link - Irwin, S.W.B. and L.T. Threadgold, 1972. Electron microscope studies of Fasciola hepatica. X. Egg formation. Exp. Parasitol., 31: 321-331.
PubMedDirect Link - Irwin, S.W.B. and J.G. Maguire, 1979. Ultrastructure of the vitelline follicles of Gorgoderina vitelliloba (Trematoda: Gorgoderidae). Int. J. Parasitol., 9: 47-53.
CrossRefDirect Link - Levran, C., L.G. Poddubnaya, R. Kuchta, M. Freeman and T. Scholz, 2007. Vitellogenesis and vitelline system in the pseudophyllidean tapeworm Paraechinophallus japonicus: Ultrastructural and cytochemical studies. Folia Parasitol., 54: 43-51.
Direct Link - Martinez-Alos, S., B. Cifrian and V. Gremigni, 1993. Ultrastructural investigation on the vitellaria of the digenean Dicrocoelium dendriticum. J. Submicrosc. Cytol. Pathol., 25: 583-590.
Direct Link - Poddubnaya, L.G., J.S. Mackiewicz, Z. Swiderski, M. Brunanaska and T. Scholz, 2005. Fine structure of egg-forming complex ducts, eggshell formation and supporting neuronal plexus in progenetic Diplocotyle olrikii (Cestoda: Spathebothriidea). Acta Parasitol., 50: 292-304.
Direct Link - Sampour, M., 2006. The study of egg shell formation in Haploporus benedenii. Bull. Eur. Assoc. Fhsh. Pathol., 25: 217-221.
Direct Link - Smyth, J.D. and J.A. Clegg, 1959. Egg-shell formation in trematodes and cestodes. Exp. Parasitol., 8: 286-323.
Direct Link - Thulin, J., 1982. Structure and function of the female reproductive ducts of the fish blood-fluke Aporocotyle simplex Odhner, 1900 (Digenea: Sanguinicolidae). Sarsia, 67: 227-248.
CrossRefDirect Link - Tulloch, G.S. and J.E. Shapiro, 1957. The ultrastructure of the vitelline cells of Haematoloechus. J. Parasitol., 43: 628-632.
Direct Link - Wells, K.E. and J.S. Cordingley, 1991. Schistosoma mansoni: Eggshell formation is regulated by pH and calcium. Exp. Parasitol., 73: 295-310.
PubMedDirect Link








