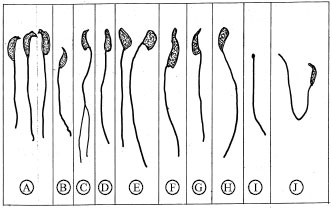
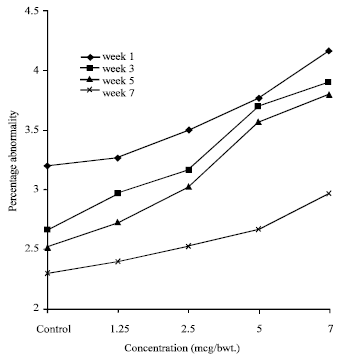
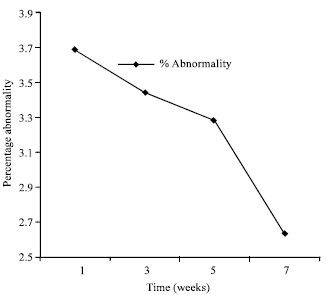
Research Article
An in vivo Evaluation of Induction of Abnormal Sperm Morphology by Ivermectin MSD (Mectizan®)
Department of Zoology, University of Lagos, Lagos, Nigeria
A.A. Mosuro
Department of Basic Sciences, Botswana College of Agriculture,Gaborone, Botswana USA
T. F. Ladipo
Department of Zoology, University of Lagos, Lagos, Nigeria