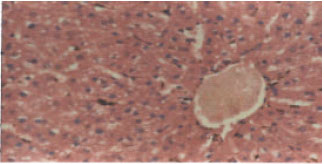
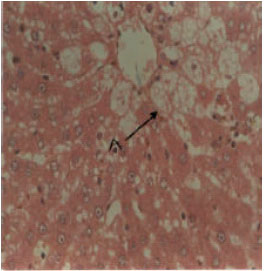
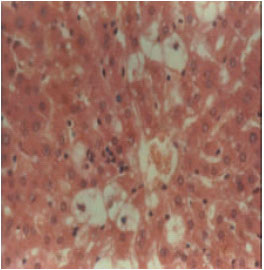
Research Article
Prevention of CCl4-Induced Liver Damage by Ginger, Garlic and Vitamin E
Department of Biochemistry, Toxicological Unit, University of Port Harcourt, P.M.B. 5323, Choba, Port Harcourt, Rivers State, Nigeria
M. O. Wegwu
Department of Biochemistry, Toxicological Unit, University of Port Harcourt, P.M.B. 5323, Choba, Port Harcourt, Rivers State, Nigeria
E. O. Ayalogu
Department of Biochemistry, Toxicological Unit, University of Port Harcourt, P.M.B. 5323, Choba, Port Harcourt, Rivers State, Nigeria