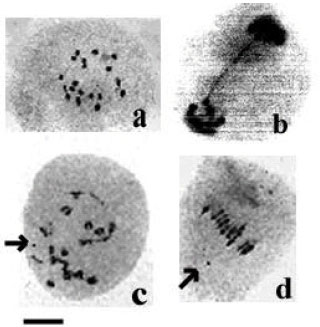
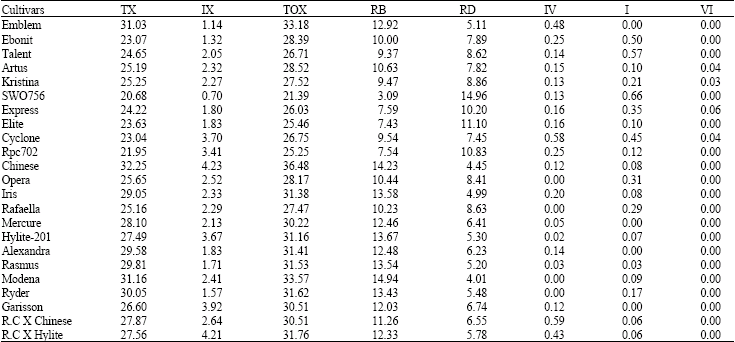
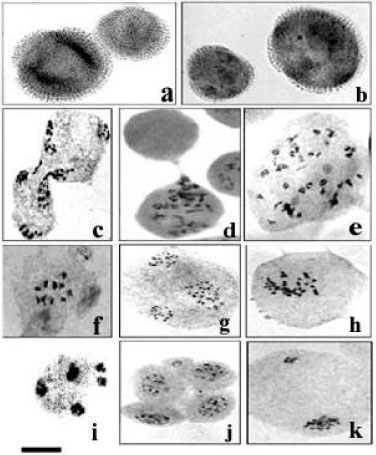
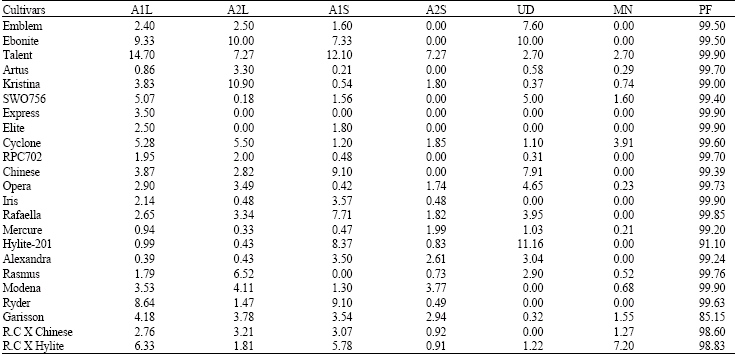
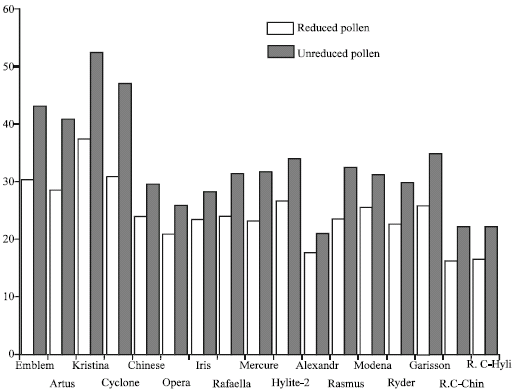
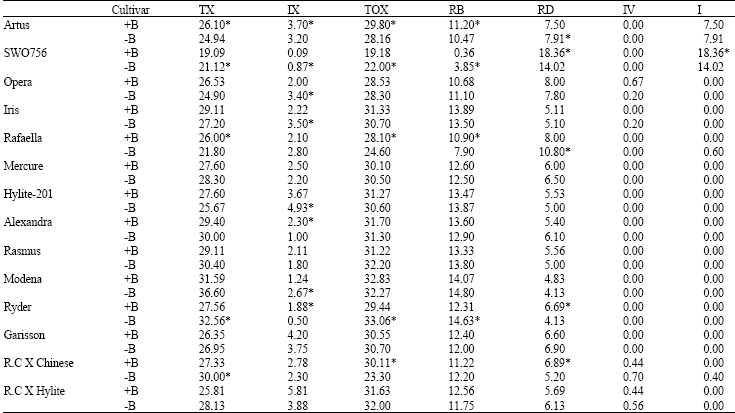
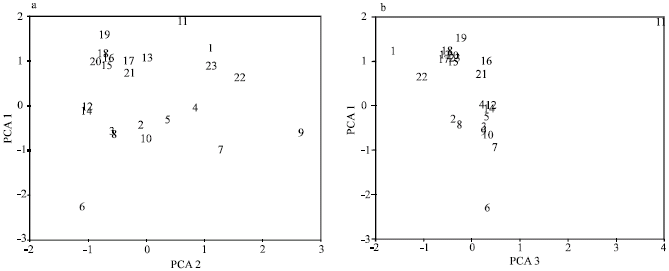
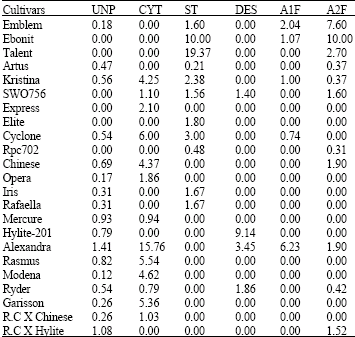
Research Article
Cytogenetic Variability in Several Oil Seed Rape Cultivars
Department of Biology, Shahid Beheshti University, Tehran, Iran
M. Sottodeh
Department of Biology, Shahid Beheshti University, Tehran, Iran
B. Akbarei
Department of Biology, Shahid Beheshti University, Tehran, Iran