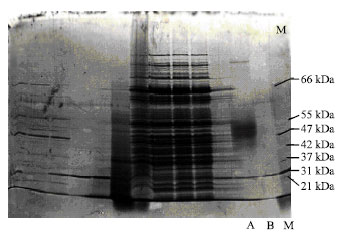
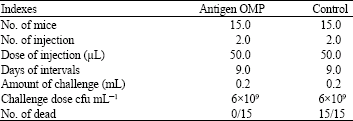
Research Article
Evaluation of Outer Membrane Proteins of Pseudomonas aeruginosa as a Protective Agent in Mice Model
Department of Medical Laboratory Sciences, Faculty of Para medicine, Tehran Medical Sciences, Tehran, Iran
Farhad Esmaily
Department of Microbiology, Razi Vaccine and Serum Research Institute, Karaj, Iran
Farhad Vaezzadeh
Department of Pathobiology, School of Public Health, Tehran University of Medical Sciences, Tehran, Iran
Hossien Dargahi
Department of Health Care Management, Faculty of Para medicine, Tehran University of Medical Sciences, Tehran, Iran