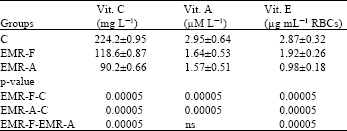
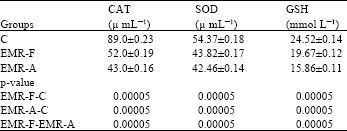
Research Article
Effects of Electromagnetic Field Produced by Mobile Phones on the Oxidant and Antioxidant Status of Rats
Department of Biophysics, Faculty of Science, Cairo University, Egypt
Ghada M. Nabil
Department of Biochemistry, National Research Center, Egypt
Atef M. M. Attia
Department of Biochemistry, National Research Center, Egypt