ABSTRACT
This investigation deals with variations in karyotype features, nuclear DNA content and nuclear volume in nine genotypes of Phaseolus vulgaris that show different adaptations to Zinc Deficiency (ZD). In addition, this study addresses the correlation between chromosome length, nuclear DNA amount and nuclear volume on one hand and the capacity of the examined genotypes to tolerate Zn deficiency on the other hand. It was found that Zn Efficiency (ZE) genotypes have the longer chromosomes, higher amounts of DNA content and larger nuclear volume than ZD genotypes.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/pjbs.2007.4230.4235
URL: https://scialert.net/abstract/?doi=pjbs.2007.4230.4235
INTRODUCTION
Genotypic variation in the capacity of plants to absorb, translocate and use mineral nutrient has been the subject of many investigations since the initial reports of Sommer and Lipman (1927) and Candler (1937). However, within the past 30 to 35 years, researchers have made great efforts to understand and exploit genetic differences of deficiency. The genetic fundamentals of nutrition among genotypes of Phaseolus vulgaris in their capacity to withstand micronutrients has been studied by Broawn and Leggett (1967), Ambler and Brown (1969), Cakmak et al. (1989), Graham and Rengel (1993), Moraghan and Grafton (1999), Westermann and Singh (2000), Singh and Westermann (2002), Forster et al. (2002), Hacisalihoglu et al. (2004), Cichy et al. (2005) and Mekki (2007).
The karyotype of Phaseolus vulgaris has been poorly characterized due to the tiny size of the chromosomes and that may explain why there is a shortage in the karyological studies of that genus (Hucl and Scoles, 1985; Zheng et al., 1991) Prior to 1996 the chromosome counts of only nine species and 4 subspecies were reported. Mercado-Ruaro and Delgado Salinas (1996, 1998) increased the number of taxa analyzed to 31 and they described the karyotype of ten wild species which represent only about 20% of those comprising the genus. They recorded a diploid number 2n = 22 in the majority of species and proposed that the basic chromosome number in the genus Phaseolus is x = 11.
Measurements of genome size in plants, estimated as nuclear DNA amount (C-value) using cytophotometric methods, indicated that the genome size is often constant for the same species (Bennet et al., 2000). In angiosperms, which include over 220000 species, C-value showed 1000-fold variations (Bennet and Leitch, 1995, 1997, 2005b; Bennet et al., 2000). However, significant infraspecific differences have been also reported in several other species (C-value database@http://www.rbgkew.org.uk). More recently infraspecific differences were also scored in Sesbania sericia and Sesbania sesban (El-Shazly et al., 2002). Much of the infraspecific variations in C-value has been attributed to genetic and environmental factors (Grime and Mowforth, 1982; Narayan, 1998). Genome size has been found to be also associated with cellular and nuclear criteria and it is generally positively correlated with chromosome size; for example in Plantago (Badr et al., 1987; Hamoud et al., 1994), in Lathyrus (Narayan, 1998) and in Sesbania (El-Shazly et al., 2002).
Constant significant intraspecific variation in nuclear DNA has been found among subspecies cultivars and genotypes of Phaseolus vulgaris (Ayonoadu, 1974; Bennett et al., 1982; Casstagnaro et al., 1990; Nagl and Treviranus, 1995). The objectives of the present study was to investigate the chromosomal variation between the nine genotypes of Phaseolus vulgaris and differentiate between those showing adaptation to zinc deficiency (ZE genotypes) and genotypes that are sensitive to (ZD). In addition, this study will address the correlation between some parameters namely chromosome length, nuclear DNA amount and nuclear volume from one hand and the capacity of the examined genotypes to tolerate Zn deficiency in soils from the other hand.
MATERIALS AND METHODS
Nine cultivars of the common bean (Phaseolus vulgaris L.) representing the major varieties cultivated in Egypt have been used in this investigation (Table 1, 2). Seeds of these genotypes were kindly supplied by the legumes section, Institute of Horticulture, Agriculture Research Center, Egypt. The study was carried out in Botany Department, Suez Canal University, Ismailia, Egypt in 1996 and 1997. The capacity of these cultivars to tolerate zinc deficiency in the soil was determined by Fekry (1999). Under Zn deficiency, visual chlorosis symptoms were first observed in leaves of the genotypes Giza-3 and Giza-4 during two seasons of study 35-40 days after sowing. After 55 days from sowing, these two genotypes developed severe Zn deficiency symptoms such as stunted growth, shortened internodes and petioles and smaller malformed leaves, which results in the classic rosette symptoms in addition to the appearance of patches on leaves. Two other genotypes; Giza-6 and Morgan showed slight Zn deficiency symptoms. The remaining five genotypes; Form, Pronco, Selendrine, Contender and Swiss-Blane appeared to be intermediate in visual Zn deficiency symptoms.
Karyotype analysis: For karyotype analysis, root tips of about 1-2 cm. were cut off from seedlings grown in Petri dishes. The roots were immediately fixed in freshly-prepared solution of 3:1 absolute ethyl alcohol: glacial acetic acid for 24 h at room temperature, washed in distilled water and then stored in 70% ethanol at 4°C until used. Cytological preparations were made using the Feulgen squash method as described by Hermann and Buban (1973). The roots were rinsed in distilled water, hydrolyzed in 1N HCl for 6 min at 60°C and thin stained in frish feulgen stain and they were made permanent by mounting in Canada balsam. Several features of the chromosomes were measured in photographic prints magnified to 3200X. These features included somatic chromosome number, total length of individual chromosome, mean chromosome length, total length of diploid chromosomes and the mean arm ratio and centromere index for each genotype. From these measurements the karyotype formula for each genotype was deduced.
Nuclear DNA content: For the determination of nuclear DNA content root tips were fixed as described above, rinsed in freshly prepared SO2 and distilled water respectively and then dehydrated and finally squashed in a drop of 45% acetic acid, freezed on CO2 ice, dehydrated in 96 and 100% ethanol respectively for 5 min and mounted in Canada balsam. According to Hesemann (1980). The DNA content of the mid telophase nuclei (2C/value) was measured, for each genotype using Flow cytometery. For these calculations, 75-100 telophase nuclei were measured in five slides. Root tip cells of Allium cepa were used as a control to calibrate the amount of nuclear DNA (Mekki, 1988).
Nuclear volume: The nuclear volume was calculated according to Dhillon (1980) from the same Feulgen stained slides used for the determination of DNA content. Two diameter measurements were made at right angles long (L) and wide (W) axis and the mean diameter (d) was calculated as VLxW and the nuclear volume was calculated by the formula :
RESULTS
The chromosome number of the nine genotypes examined is 2n = 22 confirming several previous counts. The chromosomes are small and similar in gross morphology. However, a two-fold difference in the total haploid chromosome length (TCL) and the Mean Chromosome Length (MCL) are recorded in the examined genotypes. TCL varies from 13.74 μm in the genotype Giza-3 to 29.5 μm in the genotype Morgan and increases gradually in the other genotypes as shown in Table 1. The variation in the MCL follows that recorded for TCL; it ranges between 1.25±0.13 μm in the genotype Giza-3 and 2.70±0.24 μm in the genotype Morgan (Table 1).
| Table 1: | Variation in the calculated karyotype features in the examined Phaseolus vulgaris genotypes |
 | |
| TCL: Total haploid chromosome length, MCL: Mean Chromosome Length, CI: Centromere Index, AR: Mean chromosome arm ratio, SE: Standard Error, Sm: Submetacentric; St: Subtelocentric | |
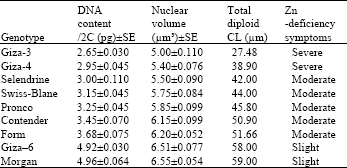
| Table 2: | Nuclear DNA content in pg, nuclear volume in μm 3 and the total diploid chromosome length in μm for the nine studied genotypes of Phaseolus vulgaris L |
 | |
The chromosomes in all of the studied genotypes consist of eight acrocentric chromosomes and three submetacentric chromosomes. However, considerable variation are found in the values of the Centromere Index (CI) and Arm Ratio (AR) among genotypes. The CI showed the highest value (23.90±2.15) in the genotype Giza -3 and it decreased gradually through the genotypes, as shown in Table 1, to reach (20.90±2.15) in the genotype Morgan. On the contrary, the arm ratio was the smallest (3.50±0.36) and it increased to 4.30±0.35 in the genotype Morgan (Table 1).
Nuclear DNA content: The nuclear DNA content of the nine studied genotypes of Phaseolus vulgaris is given (Table 2) in which the genotypes are listed. It is clearly observed that the highest amount of nuclear DNA is found in the genotype Morgan (4.96±0.064 pg), while the lowest amount of DNA per nucleus is found in the genotype Giza-3 (2.65±0.030) as increased gradually through the genotypes as shown in Table 2. The amount of nuclear DNA content ranges between 2.95±0.045 in the genotype Giza-4 to 4.92±0.030 in the genotype Giza-6.
Nuclear volume: The nuclear volume of the examined genotypes is also given in Table 2. It is clear that the nuclear volume of the two genotypes Morgan and Giza-6 is considerably high (6.55±0.054 and 6.51±0.077 μm3) respectively. In the other genotypes, the smallest nuclear volume was recorded in the genotype Giza-3 (5.00±0.110 μm3) and increased gradually through the genotypes as shown in Table 2 up to 6.20±0.052 μm3 in the genotype Form. The total length of the chromosomes, increased along the listed genotypes from 27.48 μm in the genotype Giza-3 to 59.00 μm in the genotype Morgan. Table 2 also includes the measure of the genotypes to tolerate Zn deficiency in the soil. The two genotypes Giza-3 and Giza-4 suffered severe Zn deficiency symptoms, whereas the two genotypes Giza-6 and Morgan suffered slight Zn deficiency symptoms, the other five genotypes showed moderate symptoms with regard to tolerance of Zn deficiency in the soil.
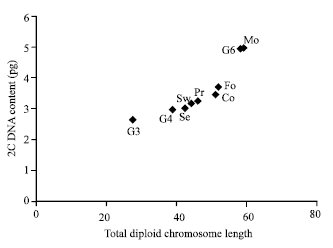 | |
| Fig. 1: | The plot of total diploid chromosome length against 2C DNA content for the nine examined Phaseolus vulgaris genotypes |
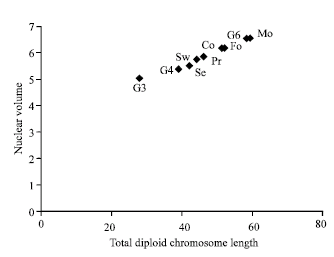 | |
| Fig. 2: | The plot of total diploid chromosome length against the nuclear volume for the nine examined Phaseolus vulgaris genotypes |
The plotting of the total diploid chromosome length against the 2C DNA nuclear content (Fig. 1), clearly distinguished the two genotypes Morgan (Mo) and Giza-6 with longer chromosomes and higher nuclear DNA amount. In this plot the genotype Giza 3 is also clearly recognized with the shortest chromosomes and the lowest 2 C DNA amount. This plot demonstrates strong positive correlation between the chromosomal length and the 2C DNA amount in the nucleus.
The plotting of the total diploid chromosome length against the nuclear volume (Fig. 2), clearly distinguished the two genotypes Morgan (Mo) and Giza-6 with longer chromosomes and larger nuclear volume and the genotype Giza 3 with the shortest chromosomes and the smallest nuclear volume. In this plot also a positive correlation is evident between the total diploid chromosome length and the nuclear volume.
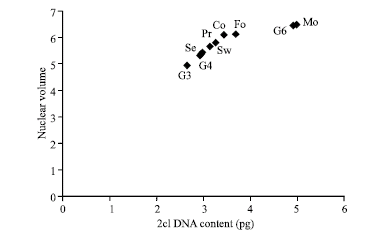 | |
| Fig. 3: | The plot of the 2C DNA nuclear amount and the nuclear volume for the nine examined Phaseolus vulgaris genotypes |
A similar positive correlation is also evident between the 2C DNA amount and the nuclear volume (Fig. 3).
DISCUSSION
The chromosome number in the nine examined genotypes of Phaseolus vulgaris L. was found to be diploid with 2n = 22 confirming earlier reports on the chromosome numbers of Phaseolus vulgaris (Karpetschenko, 1925; Mercado-Ruaro and Delgado- Salinas, 1996; 1998). The karyotype analysis revealed that the nine genotypes have eight acrocentric and three submetacentric chromosomes. These results disagree with Mercado-Ruaro and Delgado-Salinas (2000) who reported a predominance of metacentric and submetacentric chromosomes but our findings partially agree with Haq et al. (1980) who concluded that the chromosomes of Phaseolus vulgaris can arbitrarily be described with median, submedian or subterminal centromeres. These variations in the gross morphology of the chromosomes may be due to the occurrence of pericentric and paracentric inversions and translocation which have been regarded as the main factors involved in the karyotypic evolution of the genus Phaseolus by some authors (Sarbhoy, 1977, 1980; Sinha and Roy, 1979a, b).
The chromosomes of the examined genotypes are generally smaller than the size reported by Sarbhoy (1978) regarding the mean length of chromosome for Phaseolus vulgaris (3 μm). The MCL ranged from 1.25 to 2.68 μm. Present results showed that ZE genotypes have the longer chromosome and ZD genotypes have smaller chromosomes while the moderate genotypes have intermediate chromosomes.
The DNA content of the cultivated Phaseolus vulgaris has been reported by different authors as being variable, 1.56, 1.63, 1.69, 1.79 pg by Casstagnaro et al. (1990), 2.7 pg by Bennett and smith (1976) and 3.7 pg by Ayonoadu (1974) and Bennett et al. (1982). These differences may be attributable to the source of the material, the type of control used or to errors inherent in the technique (Bennett and Leitch 2005a). Present results showed variation between the nine studied genotypes ranged between 2.65 and 4.96 pg (with an average of 3.8 pg). This value may confirms the earlier results obtained by Ayonoadu (1974) and Bennett et al. (1982) and needs more investigations. Stebbins (1951) and Dolezel and Bartos (2005) pointed out that the variation in DNA content in plants is an adaptive response to different environmental gradients or growth conditions. The intraspecific variation in DNA content has been reviewed by Greilhuber (1998); to be due to non-recognized taxonomic heterogeneity or it may be due to increase or decrease in the copy sequences (Vicient et al., 1999; Zhang and Wessler, 2004). Inspite of these variations in the amount of DNA content between the nine studied genotypes we found that ZE genotypes (Morgan and Giza-6) have the highest values (4.96 and 4.92 pg) and the ZD genotypes (Giza-3 and Giza-4) have lower values (2.95 and 2.65 pg), while the moderate genotypes (Form, Contender, Pronco, Swiss-Blane and Selendrine) have intermediate values (3.68, 3.45, 3.25, 3.15, and 3 pg, respectively). These results are congruent with the findings that the longer chromosomes are found in the ZE genotypes and the shorter ones are found in ZD genotype while the other moderate genotypes were intermediate in chromosome length and nuclear DNA content.
In conclusion the results of the measurements of the nuclear volume for the nine genotypes showed also a positive correlation between DNA content and both total chromosome length and nuclear volume. This agrees with the results obtained by Mcleish and Sunderland (1969), Ayonoadu (1974) and Casstagnaro et al. (1990). Also Marcado-Ruaro and Delgado-Salinas (2000) reported that varieties with high DNA content and larger nuclei are better adapted to some environmental conditions. Sinha et al. (1995) also concluded that the higher amount of DNA may give the plants a selective advantage to cope with adverse conditions.
REFERENCES
- Ayonoadu, U.W.U., 1974. Nuclear DNA variation in phaseolus. Chromosoma, 48: 41-49.
CrossRefDirect Link - Badr, A., R. Labani and T.T. Elkington, 1986. Nuclear DNA variation in relation to cytological features of some species in the genus Plantago L. Cytologia, 52: 733-737.
Direct Link - Bennett, M.D. and J.B. Smith, 1976. Nuclear DNA amounts in angiosperms. Philos. Trans. R. Soc. B: Biol. Sci., 274: 227-274.
CrossRefPubMedDirect Link - Bennett, M.D. and Leitch, 1995. Nuclear DNA amounts in angiosperms. Ann. Bot., 76: 113-176.
CrossRefDirect Link - Bennett, M.D. and I.J. Leitch, 1997. Nuclear DNA amount in angiosperms- 583 new estimates. Ann. Bot., 80: 169-196.
Direct Link - Bennett, M.D., S. Johnston, G.L. Hodnett and H.J. Price, 2000. Allium cepa L. cultivars from four continents compared by flow cytometry show nuclear DNA constancy. Ann. Bot., 85: 351-375.
Direct Link - Bennett, M.D. and I.J. Leitch, 2005. Nuclear DNA amounts in angiosperms: Progress, problems and prospects. Ann. Bot., 95: 45-90.
CrossRefPubMedDirect Link - Cakmak, I., H. Marschner and F. Bangerth, 1989. Effect of zinc nutritional status on growth, protein metabolism and levels of indole-3-acetic acid and other phytohormones in bean (Phaseolus vulgaris L.). J. Exp. Bot., 40: 405-412.
CrossRefDirect Link - Cichy, K.A., S. Forster, K.F. Grafton and G.L. Hosfield, 2005. Inheritance of seed zinc accumulation in navy bean. Crop Sci., 45: 864-870.
Direct Link - Dolezel, J. and J. Bartos, 2005. Plant DNA flow cytometry and estimation of nuclear genome size. Ann. Bot., 95: 99-110.
CrossRefPubMedDirect Link - El-Shazly, H.H., I. El-Sheikh, M.A. El-Kholy and A. Badr, 2002. Variation in cell cycle phases duration in relation to genome size and chromosome length in some species of Sesbania. Proceedings of the 2nd International Conference on Biological Science, April 27-28, 2002, Tanta University, pp: 111-121.
- Greilhuber, J., 1998. Intraspecific variation in genome size: A critical reassessment. Ann. Bot., 82: 27-35.
Direct Link - Hacisalihoglu, G., L. Ozturk, I.R. Cakmak, M. Welch and L. Kochian, 2004. Genotypic variation in common bean in response to zinc deficiency in calcareous soil. Plant Soil, 259: 71-83.
Direct Link - Mercado-Ruaro, P. and A. Delgado-Salinas, 2000. Cytogenetic studies in Phaseolus L. (Fabaceae). Genet. Mol. Biol., 23: 985-987.
CrossRefDirect Link - Narayan, R.K.J., 1998. The role of genomic constraints upon evolutionary changes in genome size and chromosome organization. Ann. Bot., 82: 57-66.
Direct Link - Singh, S.P. and D.T. Westermann, 2002. A single dominant gene controlling resistance to soil zinc deficiency in common bean. Crop Sci., 42: 1071-1074.
Direct Link - Sommer, A.L. and C.B. Lipman, 1926. Evidence on the indispensable nature of zinc and boron for higher green plant. Plant Physiol., 1: 231-249.
CrossRef - Vicient, C.M., A. Suoniemi, K. Anamthawat-Jonsson, J. Tanskanen, A. Beharav, E. Nevo and A.H. Schulman, 1999. Retrotrasposon BARE-1 and its role in genome evolution in the genus Hordeum. Plant Cell, 11: 1769-1784.
Direct Link - Westermann, D. and S.P. Singh, 2000. Patterns of responseto zinc deficiency in dry bean of different market classes. Ann. Rpt. Bean Improv. Coop, 43: 5-6.
Direct Link - Zhang, X. and S.R. Wessler, 2004. Genome-wide comparative analysis of the transposable elements in the related species Arabidopsis thaliana and Brassica oleracea. Proc. Natl. Acad. Sci. USA., 101: 5589-5594.
Direct Link








