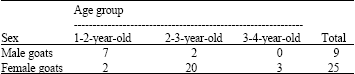Research Article
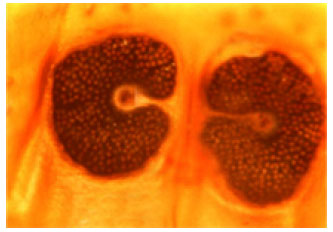
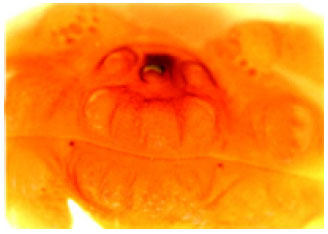
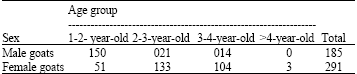
A Survey of Seasonal Infestation with Przhevalskiana Larvae in Slaughtered Goats and Sheep in South-western Iran
Department of Pathobiology,
Mehrdad Pourjafar
Department of Clinical Science, School of Veterinary Medicine, Shahrekord University,
Saeideh darabi
Faculty of Veterinary, Shahrekord University, Shahrekord, Iran
Farzaneh Assadian
Faculty of Veterinary, Shahrekord University, Shahrekord, Iran
Fariba Kahkesh
Faculty of Veterinary, Shahrekord University, Shahrekord, Iran