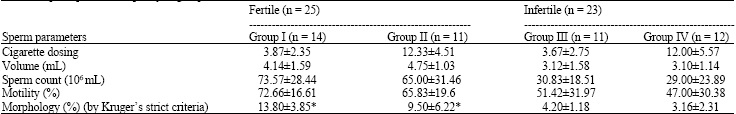
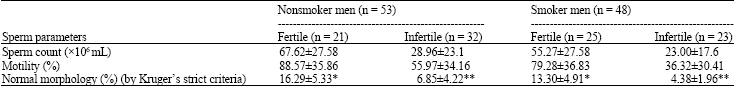Research Article
Cigarette Smoking and the Risk of Male Infertility
Department of Biology, Faculty of Basic Science, Mazandaran University, Babolsar, Iran
G. A. Jorsaraee
Department of Fertility and Infertility, Fateme Zabra Hospital, Babol University of Medical Sciences, Babol, Iran
E. Tahmasbpour Marzony
Department of Biology, Faculty of Basic Science, Mazandaran University, Babolsar, Iran