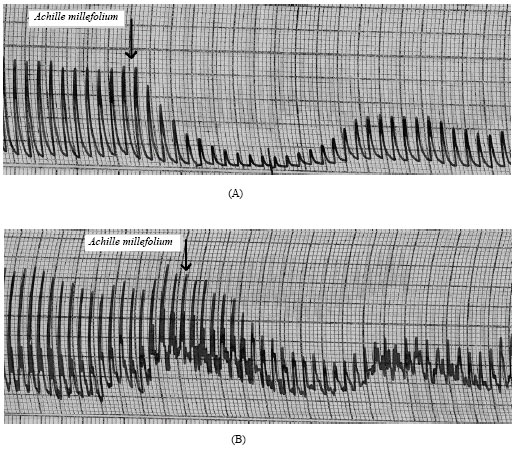
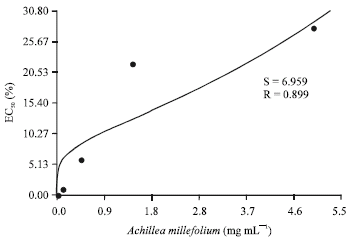
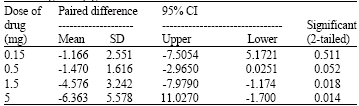
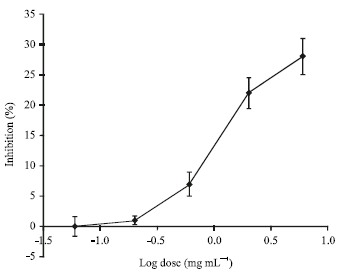
Research Article
Antimotility Effect of Hydroalcoholic Extract of Yarrow (Achillea millefolium) on the Guinea-Pig Ileum
Gastrointestinal and Liver Research Center,
Mitra Emmami Abarghoei
Department of Pharmacology, Semnan University of Medical Sciences, Semnan, Iran
Maziar Mohammad Akhavan
Department of Pharmacology, Semnan University of Medical Sciences, Semnan, Iran
Reza Ansari
Digestive Disease Research Center, Tehran University of Medical Sciences, Tehran, Iran
Abbas Ali Vafaei
Physiology Research Center, Semnan University of Medical Sciences, Semnan, Iran
Abbas Ali Taherian
Physiology Research Center, Semnan University of Medical Sciences, Semnan, Iran
Shahrokh Mousavi
Gastrointestinal and Liver Research Center,
Jafar Toussy
Gastrointestinal and Liver Research Center,