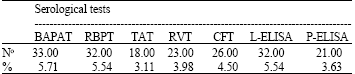
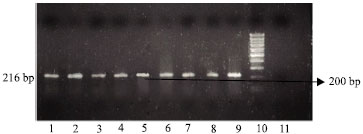
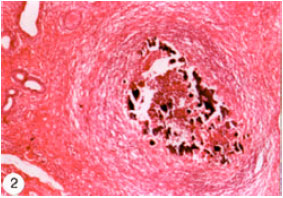
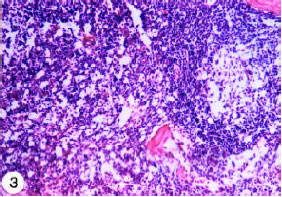
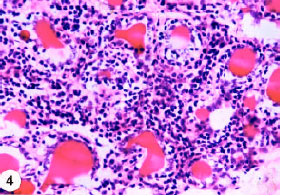
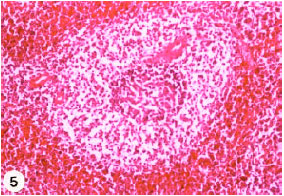
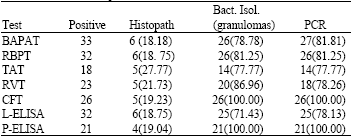
Research Article
Investigations on Brucellosis in Egyptian Baladi Does with Emphasis on Evaluation of Diagnostic Techniques
Department of Animal Reproduction and A.I. National Research Center, Dokki, Giza, Egypt
H.M. Desouky
Department of Animal Reproduction and A.I. National Research Center, Dokki, Giza, Egypt
W.M. Ahmed
Department of Animal Reproduction and A.I. National Research Center, Dokki, Giza, Egypt