Research Article
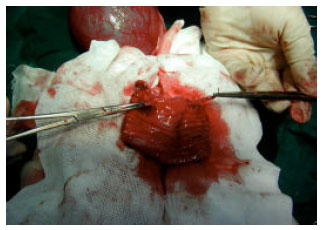
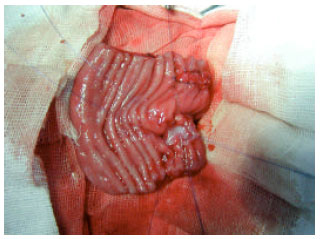
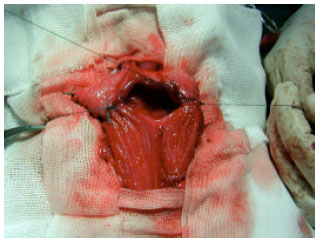
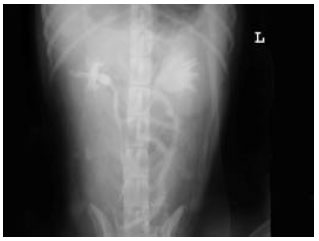
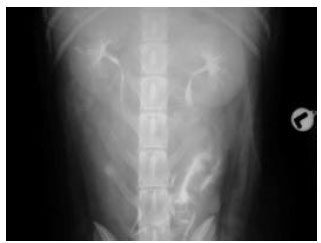
Clinical and Radiological Findings Following Continent Urinary Diversion with Colonic Segment in Dog
Department of Clinical Sciences, Faculty of Veterinary Medicine, Shahid Chamran University of Ahwaz, Iran
F. Saberi Afshar
Department of Clinical Sciences, Faculty of Veterinary Medicine, Shahid Chamran University of Ahwaz, Iran
A. R. Ghadiri
Department of Clinical Sciences, Faculty of Veterinary Medicine, Shahid Chamran University of Ahwaz, Iran