Research Article
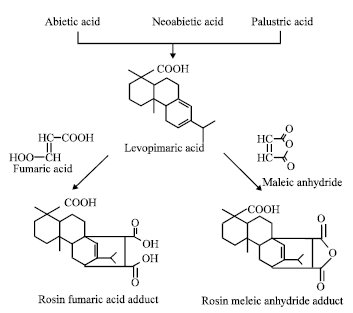
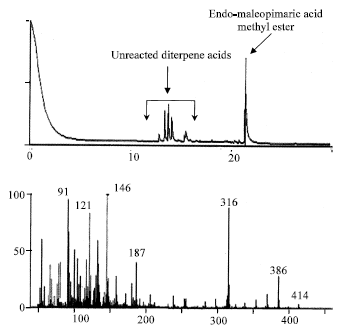
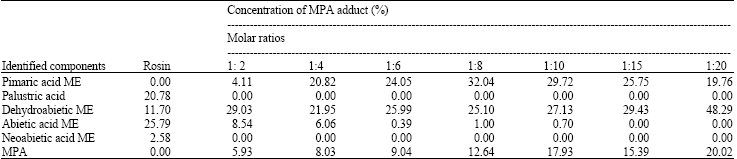
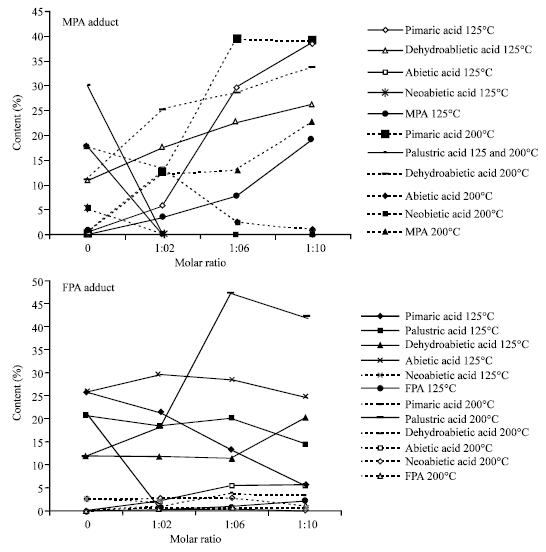
Characteristics and Chemical Composition of Maleo-Pimaric and Fumaro-Pimaric Rosins Made of Indonesian Pinus merkusii Rosin
Forest Products Research and Development Center-FORDA, Jl. Gunung Batu 5, Bogar, Indonesia
Sanro Tachibana
Faculty of Agriculture, Ehime University, 3-5-7, Tarumi, Matsuyama, Ehime, 790-8566 Japan
Djaban Tinambunan
Forest Products Research and Development Center-FORDA, Jl. Gunung Batu 5, Bogar, Indonesia