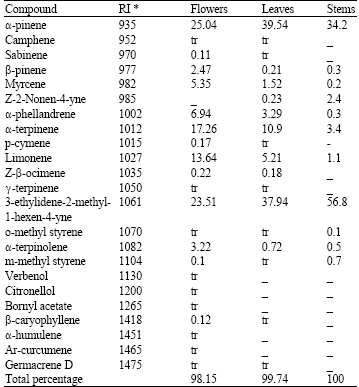
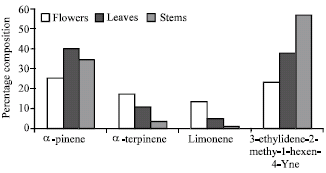
Research Article
Chemical Compositions of the Essential Oils of Stems, Leaves and Flowers of Prangos acaulis (Dc) Bornm
Department of Chemistry, Faculty of Science, University of Larestan, Iran
Hossein Hadavand Mirzaei
Department of Chemistry, Faculty of Science, University of Larestan, Iran