Research Article
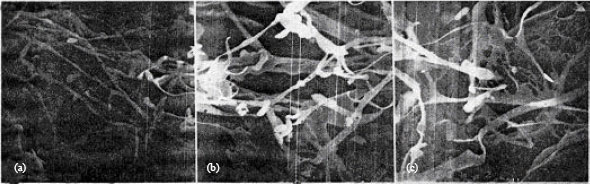
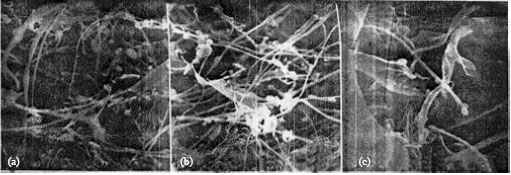
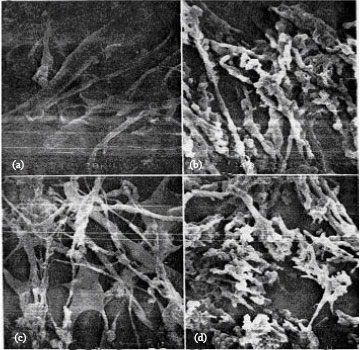
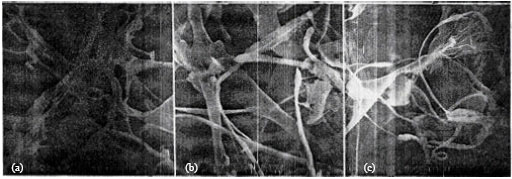
Study by Scanning Electron Microscopy of Mixture of Cereal Proteins Fractions (Maize and Wheat) from Bread Doughs Prepared at High Temperature in the Presence of Oxidants
Laboratoire de Biophysicochimie et Technologie Alimentaires. Ecole Nationale Superieure Polytechnique, Universite Marien Ngouabi, B.P. 69, Brazzaville-Congo
Antoine
Laboratoire de Biophysicochimie et Technologie Alimentaires. Ecole Nationale Superieure Polytechnique, Universite Marien Ngouabi, B.P. 69, Brazzaville-Congo