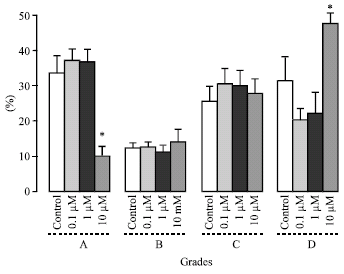
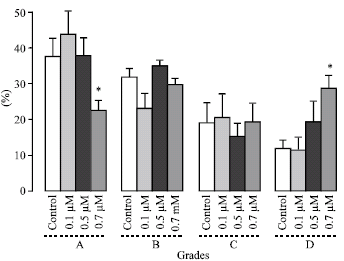
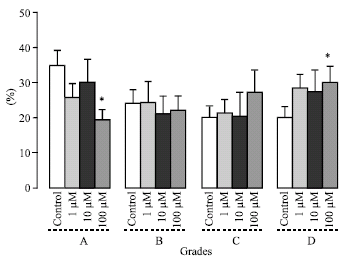
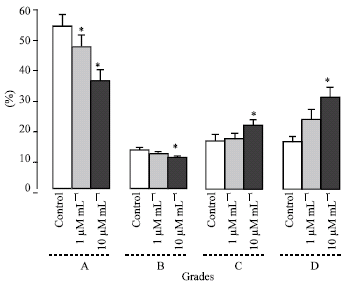
Research Article
Effect of Nitric Oxide on Ram Sperm Motility In vitro
Department of Basic Sciences, Department of Clinical Sciences, College of Veterinary Medicine, Sharekord University, Sharekord, Iran
Pejman Mirshokrai
Department of Clinical Sciences, College of Veterinary Medicine, Sharekord University, Sharekord, Iran
Abolfazl Shirazi
Department of Clinical Sciences, College of Veterinary Medicine, Sharekord University, Sharekord, Iran
Atefe Aminian
Department of Basic Sciences, Department of Clinical Sciences, College of Veterinary Medicine, Sharekord University, Sharekord, Iran