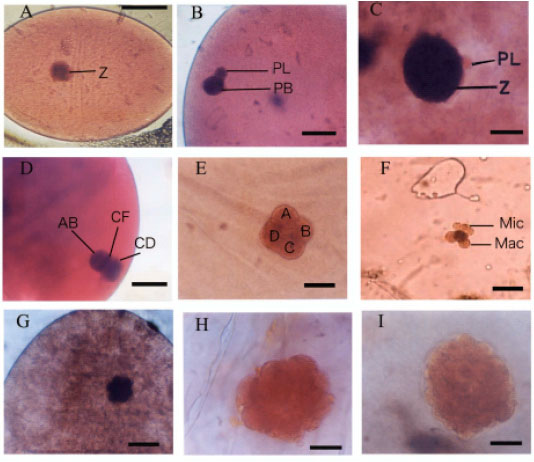
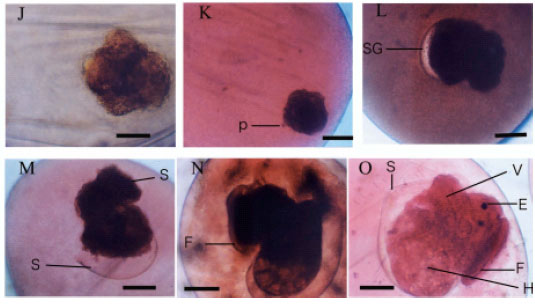
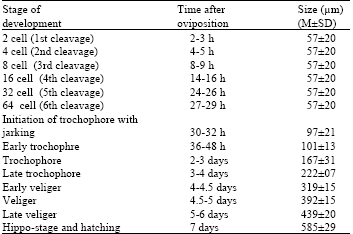
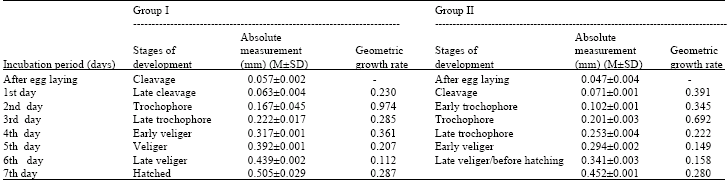
Research Article
Embryonic Developmental Ecology of Freshwater Snail Lymnaea acuminata (Lymnaeidae: Gastropoda)
Department of Chemistry, Biology and Marine Science, Faculty of Science, University of the Ryukyus, 1 Senbaru, Nishihara City, Okinawa 903-0213, Japan
Badrun Nesa
Department of Chemistry, Biology and Marine Science, Faculty of Science, University of the Ryukyus, 1 Senbaru, Nishihara City, Okinawa 903-0213, Japan
Md. Sarwar Jahan
Department of Zoology, University of Rajshahi, Bangladesh