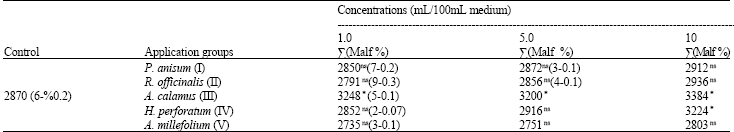
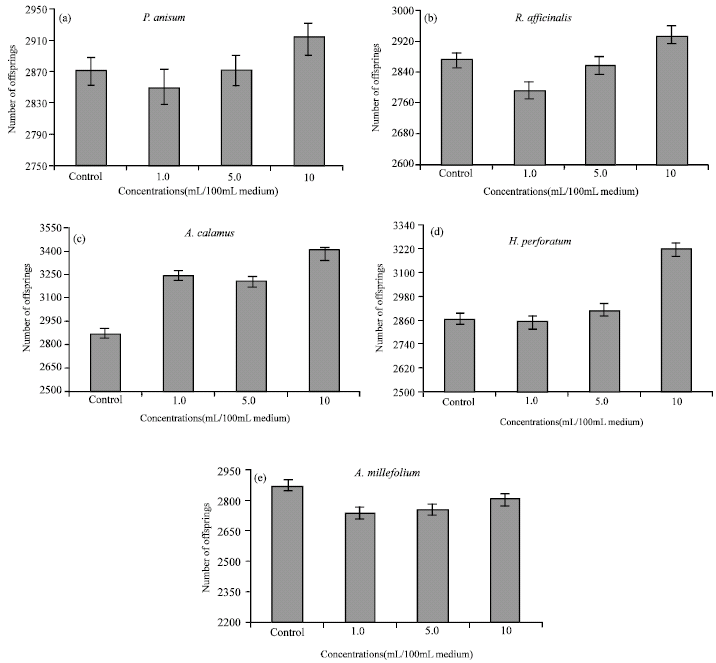
Research Article
Recovering Effects of Aqueous Extracts of Some Selected Medical Plants on the Teratogenic Effects During the Development of D. melanogaster
Department of Biology, Faculty of Science and Art, Atatiirk University, 25240/Erzurum, Turkey
Ayse Aydan Kara
Department of Biology, Faculty of Science and Art, Atatiirk University, 25240/Erzurum, Turkey
Omer Faruk Algur
Department of Biology, Faculty of Science and Art, Atatiirk University, 25240/Erzurum, Turkey
Rahmi Dumlupynar
Department of Biology, Faculty of Science and Art, Atatiirk University, 25240/Erzurum, Turkey
Mehmet Nuri Aydogan
Department of Biology, Faculty of Science and Art, Atatiirk University, 25240/Erzurum, Turkey