Research Article
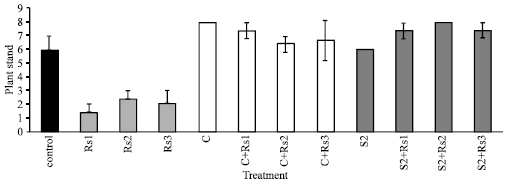
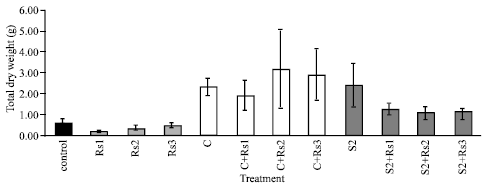
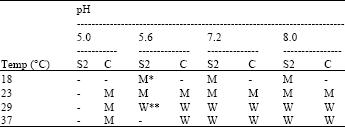
Biological Control Potential of Two Streptomyces Isolates on Rhizoctonia solani, the Causal Agent of Damping-off of Sugar Beet
Department of Genomics, Agricultural Biotechnology Research Institute of Iran, Mahdasht Road, Karaj, Iran
A.R. Hessan
Plant Diseases Lab., Plant Pests and Diseases Research Institute, Mahdasht Road, Karaj, Iran
H. Askari
Department of Plant Physiology, Biochemistry and Proteomics, Agricultural Biotechnology Research Institute of Iran
S. Aghighi
Departments of Plant Protection and Biotechnology, College of Agriculture, Shahid Bahonar University, 22 Bahman Blvd, Kerman, Iran
G.H. Shahidi Bonjar
Departments of Plant Protection and Biotechnology, College of Agriculture, Shahid Bahonar University, 22 Bahman Blvd, Kerman, Iran