ABSTRACT
Effect of temperature (20, 25 and 30°C) and photoperiod (12:12, 14:10 and 16:8 h L:D) were studied separately on Trichogramma chilonis Ishii. As the temperature increased from 20 to 30°C, fecundity increased but longevity decreased. The maximum fecundity (12.73±1.01 eggs/female/day) and adult emergence was observed at 30°C. However, the least longevity of females (6.73±0.53 days) was observed also at 30°C and the maximum (10.08±1.21) was at 20°C, which was significantly higher compared with that at 25 and 30°C. Similarly, at 14:10 h L:D fecundity (9.92±0.13 eggs/female/day) and adult emergence was significantly higher compared with 12:12 and 16:8 h L:D photoperiod regimes. There was no significant differences among different photoperiod regimes tested with respect to longevity, though it was higher at 16:8 h L:D.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/pjbs.2006.820.824
URL: https://scialert.net/abstract/?doi=pjbs.2006.820.824
INTRODUCTION
Trichogramma species are used as inundative biological control agents in more than 30 countries around the world, covering over 32 million ha of agriculture and forestry land (Li, 1994). In India, about 26 species have been reported among which Trichogramma chilonis Ishii is considered more important with regard to its distribution (Nagarkatti and Nagaraja, 1979; Ramesh and Baskaran 1996; Singh et al., 2002). Despite the accepted practice of inundative release of Trichogramma against certain pests, the degree of success obtained often varies (Ramesh and Baskaran, 1996; Prasad et al., 1999).
Rearing conditions such as food, temperature, light etc. may affect the performance of a colony being mass produced in confined situations (Hoffmann et al., 2001). Among abiotic elements, temperature and photoperiod have proved to affect biological characteristics of Trichogramma (Prasad et al., 1999; Gunie and Lauge 1997; Miura and Kobayashi, 1993; Smith and Hubbes, 1986; Harrison et al., 1985; Rounbehler and Ellington, 1973; Calvin et al., 1984).
In general, the effect of temperature on most species of Trichogramma within a defined range (9 to 30°C) is positively related to fecundity and adult emergence but inversely related to longevity (Harrison et al., 1985; Consoli and Parra, 1995; Resende and Ciociolla, 1996; Carriere and Boivin, 1997). However, there are some reports indicating that lower temperatures are preferred by some species of Trichogramma (Attaran, 2002; Miura and Kobayashi, 1993). Pak and Oatman (1982) studied T. brevicapillum and T. pretiosum at various temperatures to find an answer to their differences in natural distribution and found that the former would be successful under high temperatures and the latter under intermediate temperatures. Forsse et al. (1992) observed that under laboratory conditions lower temperatures resulted in lower level of parasitism. Haile et al. (2002) found differences among four species of Trichogramma including T. chilonis regarding their developmental time, fertility, rate of emergence and sex ratio reared at different temperatures. Pratissoli et al. (2004) demonstrated improvements in fertility life table parameters of T. pretiosum and T. acacioi from 15 to 30°C and 15 to 25°C, respectively.
Very few studies are available on the effect of photoperiod on Trichogramma and the results indicate a direct positive relationship of photoperiod with longevity and fecundity (Rounbehler and Ellington, 1973; Calvin et al., 1984; Zaslavski and Quy, 1982). One of the accessible old information on the effect of photoperiod is the studies done by Rounbehler and Ellington (1973). They found that longevity of T. semifumatum increased as the photoperiod elevated from 10 to 14 h in one strain but in another strain, maximum longevity happened at 10:14 h L:D. Zaslavski and Quy (1982) demonstrated that T. chilonis and T. evanescens had the greatest fecundity at 16:8 h L:D. Calvin et al. (1984) observed that developmental time increased in T. pretiosum under 12 and 16 h photophase, female longevity decreased significantly under 12 h photophase and fecundity was not affected by photoperiod. Garcia et al. (2000) reported that developmental time had an inverse relationship with increase in temperature and photophase in T. cordubensis.
As it has frequently been observed that different experimental conditions (temperature and photoperiod) are used by researchers to study various aspects of similar Trichogramma species, the obtained results may not be comparable or may even be controversial. From the other hand, if the experimental conditions are not uniform in studying given species of insects, then the genetical and geographical differences may not be revealed vividly. Consequently, this study was undertaken to investigate the effect of temperature and photoperiod on quality and performance of T. chilonis and to find out if some basic biological parameters of this species is related to these environmental conditions.
MATERIALS AND METHODS
The study was conducted in Department of Entomology, College of Agriculture, University of Agricultural Sciences, Bangalore, India, during 2002-2004.
Parasitoid rearing: Trichogramma chilonis Ishii culture was established by taking more than 5000 parasitized eggs of Corcyra cephalonica (Stainton) from Biological Control Research Laboratories (BCRL), Bangalore. The culture was maintained on eggs of Corcyra at 25±1°C, 65±10% RH and 16:8 h L:D.
Temperature experiment: Temperature regimes used were 20±1, 25±1and 30±1°C. Three incubators were set at either of the said temperatures. A number (>12) of newly emerged (0-24 h old) and mated females of T. chilonis were confined in homeopathic vials. Eighty to 100 fresh eggs of Corcyra were stuck on a strip of white paper (0.5x4 cm) and assigned a number, treatment code and date. Wasps were provided individually with said egg cards daily till they died. The whole set was maintained at required temperature treatment but all at 65±10% RH and 16:8 h L:D. A streak of honey was also provided inside the vials for adult feeding. Parasitized eggs were kept at the corresponding temperature and photoperiod regimes related to their treatments. After parasitized eggs turned black, blackened eggs were counted for each wasp and recorded as daily fecundity. After emergence, the number of female, male and unemerged progeny was recorded for all the treatments.
Photoperiod experiments: Photoperiod regimes were 12:12, 14:10and 16:8 h L:D. Three incubators were set at either photoperiod regime but all at 25±1 °C and 65±10% RH. For each photoperiod treatment a number of females were prepared as explained in temperature experiment and exposed to egg cards daily. Parasitized eggs were incubated in the same corresponding conditions (incubator). After 5 days, the parasitized eggs which turned black, were counted and recorded as daily fecundity for each wasp in each treatment. After emergence, female, male and unemerged progeny were noted down for all wasps in all treatments separately.
Statistical analysis: Data were subjected to GLM (Proc GLM, SAS, 1986) and means were separated by Least Significant Difference method (LSD).
RESULTS
Temperature experiments: The fecundity of T. chilonis significantly increased with increase in temperature from 20-30°C (Table 1 and Fig. 1a-c). Female progeny also had a positive relationship with the temperature. There was almost 3-fold increase in female progeny with an increase in temperature from 20 to 30°C. Male progeny and the number of unmerged were also significantly higher at 30°C compared to that at 20 and 25°C. In contrast to adult emergence and fecundity, longevity decreased with an increase in temperature from 20 to 30°C. The female longevity was significantly the highest (10.08±1.21 days) at 20°C and the lowest at 30°C. From these studies it is seen that 30°C appears to be the optimum temperature for rearing T. chilonis.
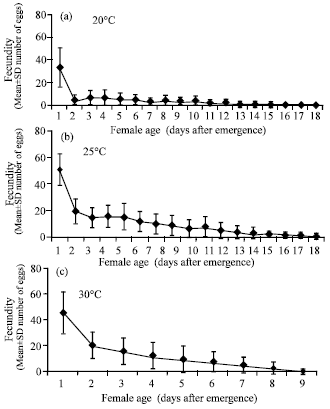 | |
| Fig. 1: | Age specific fecundity (Mean±SD number of eggs) of Trichogramma chilonis Ishii females on eggs of Corcyra cephalonica (Stainton) at 20, 25 and 30°C |
| Table 1: | Influence of rearing temperature on fecundity and adult emergence of Trichogramma chilonis Ishii females at 65±10% RH and 16:8 h L:D |
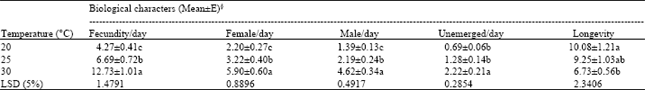 | |
| §Means with similar letter in a column are not significantly different at p<0.05 | |
| Table 2: | Influence of photoperiod on fecundity, emergence, and longevity of Trichogramma chilonis Ishii females at 25±1°C and 65±10% RH |
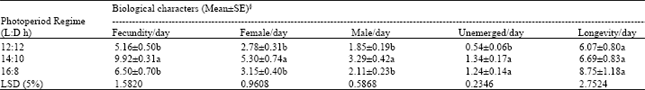 | |
| §Means with the same letter in one column are not significantly different at p<0.05 | |
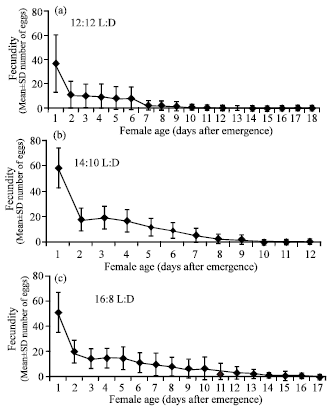 | |
| Fig. 2: | Age specific fecundity (Mean±SD number of eggs) of Trichogramma chilonis Ishii females on eggs of Corcyra cephalonica (Stainton) at 12:12, 14:10 and 16:8 h L:D |
Photoperiod experiment: T. chilonis reared at regime of 14:10 h L:D had significantly higher fecundity/day (9.92±0.31 eggs/day) compared to 12:12 or 16:8 h L:D regimes, which were on par with each other (Table 2 and Fig. 2a-c). The number of female progeny produced was also significantly higher at 14:10 h L:D regime (5.30±0.74) compared to the other two regimes. Similarly, the number of males produced were also the highest at 14:10 h L:D compared to other regimes. The photoperiod regime of 12:12 h L:D recorded the least number of unemerged/day compared to other two regimes. However, all three regimes tested did not have any influence on adult female longevity of T. chilonis (Table 2).
DISCUSSION
Temperature directly affected the developmental period of T. chilonis. Almost in all organisms there is a positive acceleration in the biological parameters when temperature increases within a thermal range (Cossins and Bowler, 1987). The effect of temperature on T. chilonis is also at par with most of earlier studies (Miura and Kobayashi, 1993; Harrison et al., 1985; Smith and Hubbes, 1986; Ramesh and Baskaran, 1996). T. chilonis has shown a kind of termophilic behaviour in other studies. Some strains of T. chilonis could tolerate up to 37±1°C (Singh et al., 2002). Calvin et al. (1984) introduced 30°C as the normal temperature for the optimum development of all stages of T. pretiosum. However, Miura and Kobayashi (1993) demonstrated that 28°C was the optimum temperature for T. chilonis developing on eggs of diamondback moth which may reveal the differences in geographical strains of T. chilonis on their sensitivity toward temperature. Moreover, Haile et al. (2002) concluded that T. chilonis from India could complete its development even at 34°C, however, the fertility decreased as the temperature increased from 25 to 34°C. All these findings are in general in agreement with the findings of the present study on the significantly better performance of T. chilonis at 30°C. Recently it was proved by Pratissoli et al. (2004) that T. pretiosum had the maximum (rm = 0.47) and doubling time (λ = 1.61) at 30°C but those of T. acacioci occurred at 25°C. Therefore, with the observation recorded during this investigation, the studied strain of T. chilonis had significantly higher fecundity (≥85 eggs/female/life time) at 30°C, although longevity decreased compared with that at 25°C, which gave rise to lower fecundity (≤60 eggs/female/life time) despite of longer life span.
The photoperiod significantly affected all the biological parameters studied in T. chilonis. However, there are very few studies available on the effect of photoperiod on life histories of Trichogramma. Most reports on this aspect emphasize on the effect of photoperiod on diapause initiation in Trichogramma (Bennemaison, 1972; Zaslavski and Umarova, 1990) and on the emergence rhythm (Zinovjeva et al., 1996). Zaslavski and Quy (1982) reported that fecundity of T. chilonis and T. evanescens increases in long day photoperiod regimes. It was proved by the present study that a 14:10 h L:D resulted in increased fecundity and adult emergence in T. chilonis, although, longevity increased with the increase in light duration (16 h) but it was not significantly different with 14 or 12 h light regime. This is not in conformity with the findings of Zaslavski and Quy (1982) mentioned before. However, even in their studies, long day did not evoke increase in fecundity of a third species named T. euproctidis. According to the results of Calvin et al. (1984) the developmental rate (mean±SE) of T. pretiosum increased at 12:12 (10.53±0.26) and 16:8 (9.15±0.10) h L:D but decreased at 14:10 (7.80±0.23) h L:D and fecundity was not affected by photoperiod. All these interesting findings prove that effectiveness of this environmental parameter on different species of Trichogramma differs greatly.
It can be concluded that as T. chilonis strain studied was taken from BCRL, Bangalore, reared in condition near to local natural environment with respect to day length and temperature, had experienced almost a 14:10 h day and night length, it may have been adopted for shorter day than 16 h light and higher temperatures. Therefore, using 25°C selected based on previous recommendations for evaluating photoperiod effects even in this investigation caused differences in measured parameters. However, it can be suggested that T. chilonis preferred 30°C and 14:10 h L:D which can be used in lab studies as well as mass rearing conditions. Compiling more information on the matter can lead to standardizing conditions in studying this wasp and making mass rearing systems more economic by saving time and money (shortening egg to adult duration and increasing adult products) as well as guarantying the quality and performance.
ACKNOWLEDGMENTS
I am extremely thankful to Biological Control Research Laboratories, Bangalore for providing initial stock of parasitoid and host eggs during the course of experiments. I thank Dr. C.A. Viraktamath, Dr. A.R.V. Kumar, Dr. K.N. Ganeshaiah and Dr. D. Teerthaprasad, University of Agricultural Sciences, GKVK, Bangalore for their guidance and encouragement during the study. I also thank Dr K.P. Jayanth, Biocontrol Research Laboratory, Bangalore for guidance and facilities provided. The grant of the Ministry of Agriculture and Ministry of Science, Research and Technology, IR Iran, for my Ph.D course in UAS, Bangalore is appreciated.








