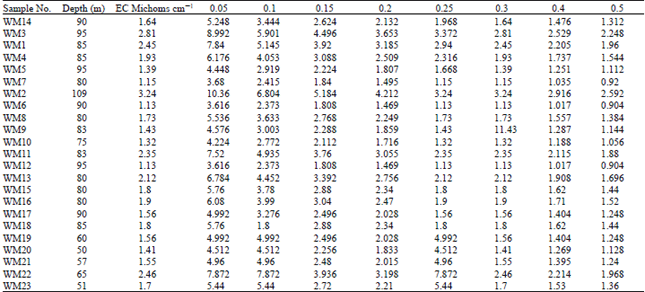
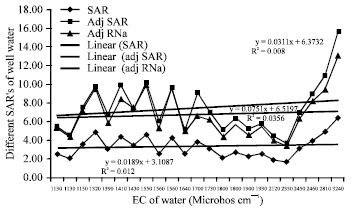
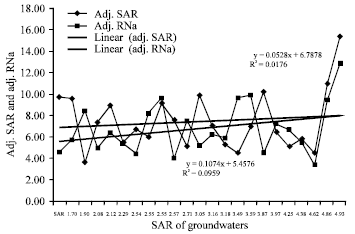
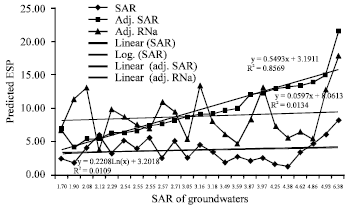
Research Article
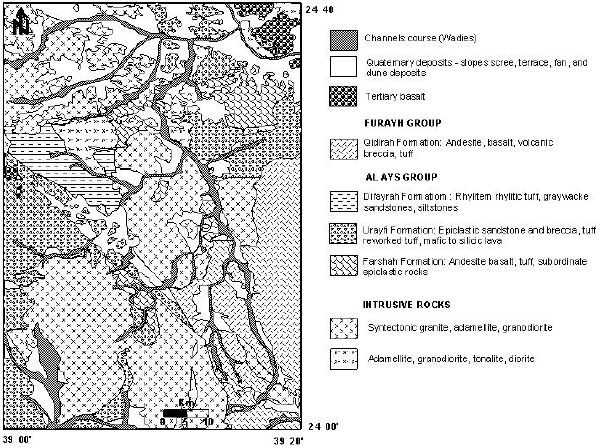
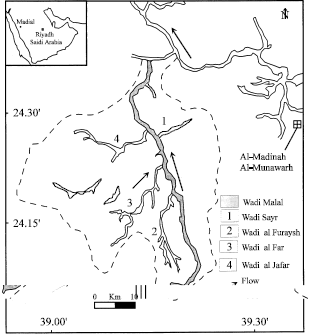
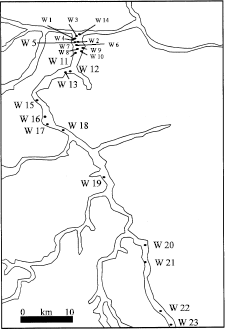
Evaluation of Groundwater Quality and its Recharge by Isotopes and Solute Chemistry in Wadi Malal, Al-Madinah Al-Munawarah, Saudi Arabia
Natural Resources and Environment Research Institute (NRERI), King Abdulaziz City for Sciences and Technology (KACST), P.O. Box 6086 Riyadh 11442, Kingdom of Saudi Arabia
Ghulam Hussain
Natural Resources and Environment Research Institute (NRERI), King Abdulaziz City for Sciences and Technology (KACST), P.O. Box 6086 Riyadh 11442, Kingdom of Saudi Arabia
M. Mujtaba Khan
Natural Resources and Environment Research Institute (NRERI), King Abdulaziz City for Sciences and Technology (KACST), P.O. Box 6086 Riyadh 11442, Kingdom of Saudi Arabia
Mohammaed A. Moallim
Natural Resources and Environment Research Institute (NRERI), King Abdulaziz City for Sciences and Technology (KACST), P.O. Box 6086 Riyadh 11442, Kingdom of Saudi Arabia
Ibrahim A. Al-Sagaby
Natural Resources and Environment Research Institute (NRERI), King Abdulaziz City for Sciences and Technology (KACST), P.O. Box 6086 Riyadh 11442, Kingdom of Saudi Arabia