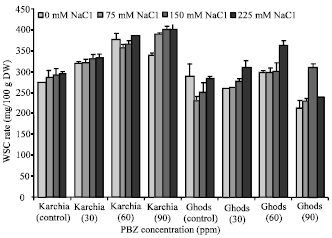
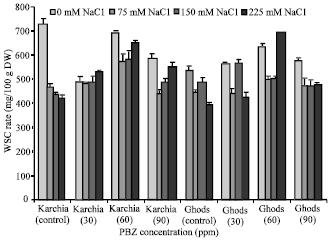
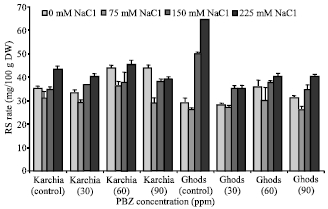
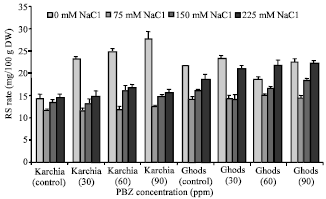
Research Article
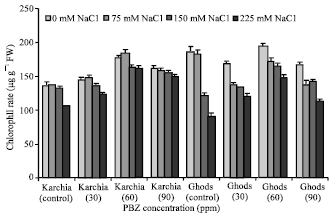
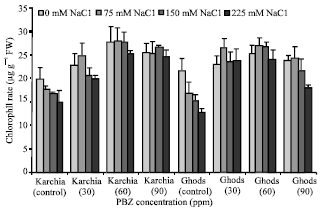
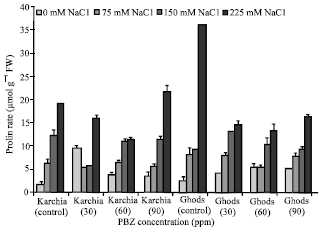
The Effects of Salt Stress and Paclobutrazol on Some Physiological Parameters of Two Salt-tolerant and Salt-sensitive Cultivars of Wheat
Departments of Biology, Faculty of Science, Azzahra University, Tehran, Iran
Khadijeh Kiarostami
Departments of Biology, Faculty of Science, Azzahra University, Tehran, Iran
Shekoofeh Enteshari
Department of Agriculture University of Valiasr, Rafsanjan, Iran
Azra Saboora
Departments of Biology, Faculty of Science, Azzahra University, Tehran, Iran