ABSTRACT
Fungal as well as algal flora inhabiting different types of hydrophytes (floating, submerged and emerged) in case of healthy and unhealthy ones and from different habitats represent different aquatic ecotypes in middle Egypt area (River Nile, channels, drains and pools) were studied. The relations between fungal and algal flora were studied. Some in vivo experiments were done to analyze and confirm the in vitro observations. In addition to complete analysis of ecological parameters which may affect the abilities of microflora to colonize and inhabitant these hydrophytes, e.g. complete physical and chemical analysis of water of investigated sites, also phytochemical investigations of studied hydrophytes were conducted.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/pjbs.2003.610.614
URL: https://scialert.net/abstract/?doi=pjbs.2003.610.614
INTRODUCTION
The protein component of the aquaculture diet is the single most expensive portion and important dietary nutrient. Fish meal remains the major dietary protein source ranging between 20-60% of the fish feed (Kaushik, 1989; Tacon and Jackson, 1985) but escalating cost, uncertainty in availability and lesser quantity has necessitated the use of other animal and plant protein sources to reduce the feed cost without compromising growth. Therefore, efforts are now being directed in different parts of the world to find alternate protein sources of good quality, which ideally are less expensive and readily available as substitutes for the expensive fish meal component in the practical diets. In this respect due to low cost and market availability soybean has been identified universally as most attractive vegetable protein source for animal feeds (Lim and Akiyama, 1989).
Wong et al. (1996), prepared a successful diet containing soybean residue with beef-liver mixture for common carp. Defatted soybean meal has universal acceptance both qualitatively and quantitatively. Soybean meal has a favorable amino acid profile as compared to other plant protein sources, is consistently available, cost effective and is reported to be palatable to most species of fish (Akiyama, 1988).
Studies on utilization of soybean meal in fish feeds have begun recently. Most of the work pertains to Trout, Salmon, Channel catfish and Tilapia, by several workers. Successful use of soybean and its products in fish feeds for the purpose of partial replacement of protein in diets of Indian fishes have been demonstrated by Chakrabarthy et al. (1973); Bhat et al. (1986); Keshabvappa, (1990) and Devaraj, (1991). Xie et al. (2001) tried a substitution experiment on gibel carp by using soybean cake, potato protein concentrate, peanut cake, cotton seed cake and rapeseed cake.
Little or no information is available on the utilization of sunflower meal in fish diet. Rab (1993) reported the partial utilization of sunflower meal in Trout feed with considerable success and better growth rate. The current experiment was conducted to determine the effect of replacement of fish meal on growth of common carp and to find out the nutritive value of soybean and sunflower meal in the feed, in order to minimize the dependence upon fishmeal as protein source and to reduce the cost of feed.
MATERIALS AND METHODS
An eight week trial was conducted in the wet labs of Aquaculture and Fisheries Institute, National Agricultural Research Center (NARC) Islamabad.
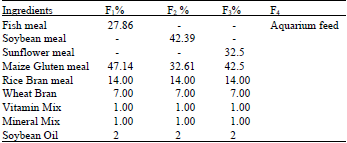
| Table 1(a): | Experimental feed compositions for Cyprinus carpio |
 | |
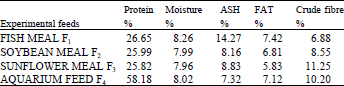
| Table 1(b): | Proximate analysis of the experimental feeds for Cyprinus carpio |
 | |
In that 200 fingerlings of common carp (Cyprinus carpio) were collected by drag net from Mein Hart Dam and the Fishponds at Aquaculture and Fisheries (NARC), Islamabad. The fish was kept in the cemented raceways and acclimatized for fifteen days on the feed supplement containing rice bran and rapeseed, in order to habituate them for artificial feeding.
Eight glass aquaria (2`x1.5`x1`) were filled with 40 L of water and aerated properly through an air pump model SSP-40 GJ-L. Out of 200 fish, 80 fingerlings of approximately same weight and length were selected and distributed randomly among the aquaria.
Three iso-nitrogenous feeds were formulated, using Pearson method, with 26% crude protein level. Fishmeal based diet was designated as F1, soybean meal diet as F2, sunflower meal as F3 where as aquarium feed (used for ornamental fish feeding) was designated as F4. The level of ingredients in each test diet was calculated on the bases of level of replacement of fishmeal (Table 1a). The dry ingredients of the feed were grinded with the help of electric grinder, weighed and mixed together in the required quantity. Mineral premix, vitamin powder and required quantity of soybean oil were also added to the feed mixture. Proximate analysis of the experimental feeds was also done for comparison (Table 1b).
Water quality parameters viz. pH, Dissolved Oxygen content, CO2, total alkalinity, total hardness and temperature were also analyzed weekly through Hatch Kit.
Final weights of the stock were taken at the end of the experiment. From the data, Food Consumption Ratio (FCR) and Specific Growth Rate (SGR) were calculated. The data was then subjected to statistical analysis (ANOVA, DMR Test, Regression analysis), using MSTAT, for growth comparisons and calculation of cost effectiveness of the four test diets.
RESULTS AND DISCUSSION
The results obtained for weight gain showed that fish fed on fish meal (F1) and aquarium feed (F4) both gained 2.82 g weight in the period of eight weeks, while Sunflower meal fed fish (F3) gained 2.01 g and Soybean fed (F2) gained 1.56 grams respectively (Table 2).
Statistical analysis for growth showed that specific growth rate of F4 was highly significant as compared with F1, because of the lesser FCR of F4 (Table 3, Fig. 1). F2 and F3 showed less significant results for growth. This may be because F1 and F2 had animal protein source as compared to F2 and F3 having plant protein source (Mc Coy, 1990 and Keshabvappa et al., 1990).
| Table 2: | Weight Gain of Cyprinus carpio with different treatments |
 | |
| Table 3: | Statistical Results for comparison of Growth in different treatments |
 | |
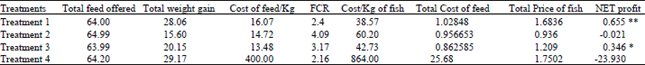
| Table 4: | Analysis in terms of Cost and Profit for different treatments |
 | |
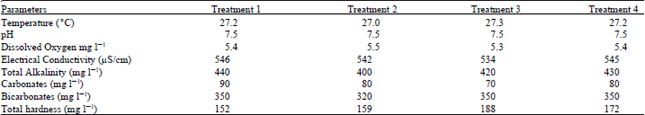
| Table 5: | Physico-chemical factors of water in Treatments 1, 2, 3 and 4 |
 | |
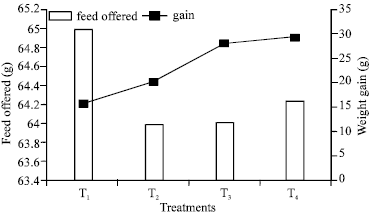 | |
| Fig. 1: | Growth performance of Cyprinus carpio for different treatments |
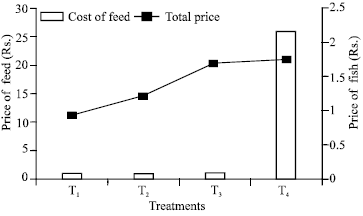 | |
| Fig. 2: | Relationship between profit and cost of Cyprinus carpio for different treatments |
Sunflower meal was proved better than the soybean meal, which may be because of low sulpher content (NRC, 1974), amino acid imbalance (Andrews and Page, 1974), anti nutritional factors other than trypsin inhibitors present in soybean meal (Wilson and Poe, 1985), low energy content (Viola et al., 1982) or lack of Phosphorus (Kim and Oh, 1985). Which may make it unpalatable and cause growth reductions may reduce or delay feed intake (Tacon et al., 1983). The better performance of Sunflower meal may be due to the absence of some of these growth inhibitors.
Contrary to the interpretation that aquarium feed is better than fish meal, when statistical analysis for cost effectiveness was done, aquarium feed proved highly non significant. In this analysis fishmeal came out to be highly significant while Soybean meal was also non-significant and Sunflower meal was significant (Table 4, Fig. 2).
Results of water quality parameters like temperature, pH, alkalinity, salinity, dissolved Oxygen, dissolved CO2, NH3 and nitrates are presented in Table 5. Since the difference between the physico-chemical factors of all aquaria was non-significant they were not supposed to affect the growth rate.
The experiment concludes that the fishmeal could not be replaced totally with plant products; however, partial replacement can be done using sunflower meal to reduce the cost without affecting growth rate.
REFERENCES
- Bisaria, V.S. and T.K. Ghose, 1981. Biodegradation of cellulosic materials: Substrates, microorganisms, enzymes and products. Enzyme Microb. Technol., 3: 90-104.
CrossRef - Cattaneo, A., G. Galanti, S. Gentinetta and S. Romo, 1998. Epiphytic algae and macroinvertebrates on submerged and floating‐leaved macrophytes in an Italian lake. Freshwater Biol., 39: 725-740.
CrossRefDirect Link - Golterman, H.L., R.S. Climo and M.A.A. Ohnstad, 1978. Methods for Physical and Chemical Analysis of Fresh Waters. 2nd Edn., Blackwell Scientific Publications, Oxford, Pages: 213.
Direct Link - Moubasher, A.H., H.M. El-Sharouny and R.A. Dadran, 1990. Fungi associated with cellulose exposed to Nile water refuses of Qena city Upper Egypt. Egypt. J. Bot., 33: 1-14.
Direct Link








