Research Article
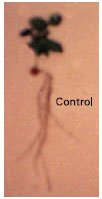
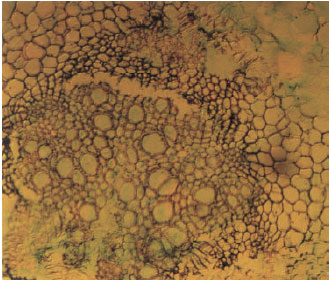
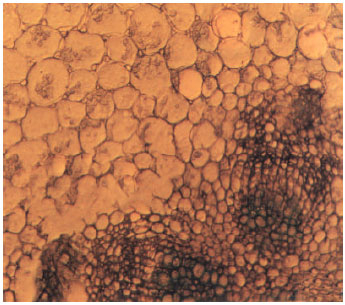
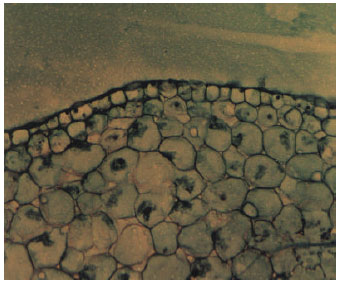
Study of the External and Internal Morphology of Pisum sativum L., with Growth Hormones i.e., Indole-3-acetic Acid and Kinetin and Heavy Metal i.e., Lead Nitrate
Department of Botany, University of the Punjab, Quaid-i-Azam Campus, Lahore 54590, Pakistan
Saima Rasheed
Department of Botany, University of the Punjab, Quaid-i-Azam Campus, Lahore 54590, Pakistan