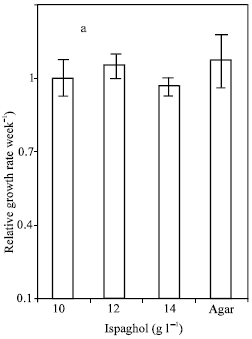
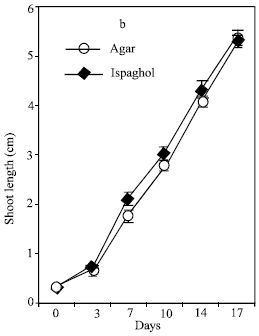
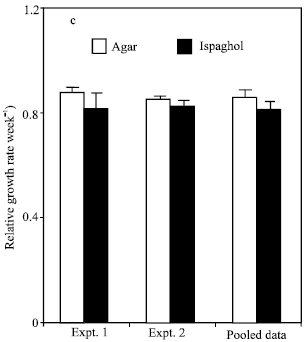
Research Article
Cost Effective Micropropagation Technology for Potatoes
Institute of Biotechnology and Genetic Engineering, NWFP Agricultural University, Peshawar, Pakistan
Safdar Hussain Shah
Institute of Biotechnology and Genetic Engineering, NWFP Agricultural University, Peshawar, Pakistan
Zahoor A. Swati
Institute of Biotechnology and Genetic Engineering, NWFP Agricultural University, Peshawar, Pakistan
Zahid Hussain
Institute of Biotechnology and Genetic Engineering, NWFP Agricultural University, Peshawar, Pakistan